UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
FORM 8-K
CURRENT REPORT
Pursuant to Section 13 or 15(d)
of the Securities Exchange Act of 1934
Date of report (Date of earliest event reported): March 13, 2026
(Exact name of Registrant as Specified in Its Charter)
(State or Other Jurisdiction of Incorporation) | (Commission File Number) | (IRS Employer Identification No.) | ||||||
| (Address of Principal Executive Offices) | (Zip Code) | ||||
Registrant’s Telephone Number, Including Area Code: (617 ) 344-4190
(Former Name or Former Address, if Changed Since Last Report)
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions (see General Instructions A.2. below):
| Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) | |||||
| Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) | |||||
| Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) | |||||
| Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) | |||||
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class | Trading Symbol(s) | Name of each exchange on which registered | ||||||||||||
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
Emerging growth company x
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. o
Item 1.01 Entry into a Material Definitive Agreement.
Securities Purchase Agreement
On March 13, 2026, Acumen Pharmaceuticals, Inc. (the “Company”) entered into a Securities Purchase Agreement (the “Purchase Agreement”) with certain institutional accredited investors named therein (each, an “Investor” and collectively, the “Investors”), pursuant to which the Company agreed to sell to the Investors, in a private placement (the “Private Placement”), 10,833,331 shares (the “Shares”) of the Company’s common stock, par value $0.0001 per share (the “Common Stock”), at an offering price of $3.30 per Share. The gross proceeds of the Private Placement are expected to be approximately $35.75 million, before deducting applicable fees and other expenses.
The Private Placement is expected to close on March 16, 2026, subject to the satisfaction of customary closing conditions. The Company intends to use the net proceeds from the Private Placement to primarily support its Enhanced Brain Delivery (EBD) program, including ongoing preclinical development work to support the nomination of a lead clinical candidate molecule, and for working capital and other general corporate purposes.
The Purchase Agreement contains customary representations, warranties and agreements by the Company, customary conditions to closing, indemnification obligations of the Company, other obligations of the parties and termination provisions. Pursuant to the Purchase Agreement, the Company has agreed to certain restrictions on the issuance and sale of shares of the Company’s securities for a period beginning on the date of the Purchase Agreement until the earlier of (i) the sixty days after the closing date of the Private Placement and (ii) the business day immediately following the date the resale registration statement filed pursuant to the Registration Rights Agreement (as defined below) becomes effective, subject to certain exceptions.
The Shares to be issued pursuant to the Purchase Agreement have not been registered under the Securities Act of 1933, as amended (the “Securities Act”), or any state securities laws and will be issued pursuant to the exemption from registration provided for under Section 4(a)(2) of the Securities Act. The Company relied on this exemption from registration based in part on representations made by the Investors. The Shares may not be offered or sold in the United States absent registration or an applicable exemption from registration requirements. Neither this Current Report on Form 8-K, nor any exhibit attached hereto, is an offer to sell or the solicitation of an offer to buy the Shares described herein.
Registration Rights Agreement
In connection with the Private Placement, the Company and the Investors entered into a Registration Rights Agreement, dated March 13, 2026 (the “Registration Rights Agreement”), providing for the registration for resale of the Shares pursuant to a registration statement (the “Registration Statement”) to be filed with the Securities and Exchange Commission (the “SEC”) no later than two business days after the date on which the Company files with the SEC its Annual Report on Form 10-K for the fiscal year ended December 31, 2025. The Company has agreed to use reasonable best efforts to cause the Registration Statement to be declared effective as soon as practicable, but in any event no later than the earlier of (i) the 75th calendar day following the initial filing of the Registration Statement if the SEC notifies the Company it will “review” the Registration Statement and (ii) the fifth business day after the date the Company is notified (orally or in writing, whichever is earlier) by the SEC that the Registration Statement will not be “reviewed” or will not be subject to further review, and to keep the Registration Statement continuously effective from the date on which the SEC declares the Registration Statement to be effective until (i) the date on which the Investors shall have resold all the Registrable Securities (as such term is defined in the Registration Rights Agreement) covered thereby, and (ii) the date on which the Registrable Securities may be resold by the Investors without registration and without regard to any volume or manner-of-sale limitations by reason of Rule 144 as promulgated by the SEC under the Securities Act (“Rule 144”), without the requirement for the Company to be in compliance with the current public information requirement under Rule 144 under the Securities Act or any other rule of similar effect.
The Company has granted the Investors customary indemnification rights in connection with the Registration Rights Agreement. The Investors have also granted the Company customary indemnification rights in connection with the Registration Rights Agreement.
The foregoing descriptions of the Purchase Agreement and the Registration Rights Agreement are not complete and are qualified in their entirety by reference to the full text of the Purchase Agreement and Registration Rights Agreement, which are filed as Exhibits 10.1 and 10.2, respectively, to this Current Report on Form 8-K and are incorporated by reference herein.
Item 3.02 Unregistered Sales of Equity Securities.
The information contained in Item 1.01 of this Current Report on Form 8-K is incorporated by reference into this Item 3.02.
Neither this Current Report on Form 8-K nor any exhibit attached hereto is an offer to sell or the solicitation of an offer to buy any securities of the Company.
Item 7.01 Regulation FD Disclosure.
On March 16, 2026, the Company issued a press release announcing the Private Placement and certain preclinical data, including in vitro, in vivo and non-human primate study results, that support development candidates in the Company’s transferrin-receptor targeting EBD program. The press release is attached as Exhibit 99.1 to this Current Report on Form 8-K.
On March 16, 2026, the Company posted an updated corporate presentation to its website at https://investors.acumenpharm.com/news-events/presentations, which the Company may use from time to time in communications or conferences. The corporate presentation was updated to include a summary of the preclinical non-human primate data from the Company’s EBD program. A copy of the presentation is attached as Exhibit 99.2 to this Current Report on Form 8-K.
The information in this Item 7.01, including Exhibit 99.1 and Exhibit 99.2 attached hereto, shall not be deemed “filed” for purposes of Section 18 of the Securities Exchange Act of 1934, as amended, or otherwise subject to the liabilities of that section, nor shall it be deemed incorporated by reference in any filing under the Securities Act of 1933, as amended, except as expressly set forth by specific reference in such filing. The Company’s submission of this Report shall not be deemed an admission as to the materiality of any information required to be disclosed solely to satisfy the requirements of Regulation FD.
Note Regarding Forward-Looking Statements
This Current Report on Form 8-K includes forward-looking statements. These forward-looking statements generally can be identified by the use of words such as “anticipate,” “expect,” “plan,” “could,” “may,” “will,” “believe,” “estimate,” “forecast,” “goal,” “project,” and other words of similar meaning. These forward-looking statements address various matters, including the expected closing date of the Private Placement and the expected use of proceeds from the Private Placement. Each forward-looking statement contained in this Current Report on Form 8-K is subject to risks and uncertainties that could cause actual results to differ materially from those expressed or implied by such statement. Applicable risks and uncertainties include, among others, the risk that the conditions to closing of the Private Placement are not satisfied; that the development and commercialization of our product candidates may take longer or cost more than planned, including as a result of conducting additional studies or our decisions regarding execution of such commercialization; that raising additional capital may cause dilution to our stockholders; that our product candidates may not receive regulatory approval, become commercially successful products, or result in new treatment options being offered to patients; and the risks identified under the heading “Risk Factors” in our Annual Report on Form 10-K for the fiscal year ended December 31, 2024, and filed with the SEC, as well as the other information we file with the SEC. We caution investors not to place considerable reliance on the forward-looking statements contained in this presentation. You are encouraged to read our filings with the SEC for a discussion of these and other risks and uncertainties. The forward-looking statements in this Current Report on Form 8-K speak only as of the date hereof, and we undertake no obligation to update or revise any of these statements. Our business is subject to substantial risks and uncertainties, including those referenced above. Investors, potential investors, and others should give careful consideration to these risks and uncertainties.
Item 9.01 Financial Statements and Exhibits.
| Exhibit No. | Description | |||||||
| 10.1* | ||||||||
| 10.2 | ||||||||
| 99.1 | ||||||||
| 99.2 | ||||||||
| 104 | Cover Page Interactive Data File (embedded within the Inline XBRL document) | |||||||
* Certain schedules, exhibits and similar attachments have been omitted pursuant to Item 601(a)(5) of Regulation S-K. The Company will provide a copy of such omitted materials to the SEC or its staff upon request.
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
| Acumen Pharmaceuticals, Inc. | ||||||||
| Dated: March 16, 2026 | By: | /s/ Derek Meisner | ||||||
Derek Meisner Chief Legal Officer | ||||||||
Exhibit 10.1
[Execution Version]
SECURITIES PURCHASE AGREEMENT
This SECURITIES PURCHASE AGREEMENT (this “Agreement”) is dated as of March 13, 2026, by and among Acumen Pharmaceuticals, Inc., a Delaware corporation (the “Company”), and each of the entities listed on Exhibit A attached to this Agreement (each, an “Investor” and together, the “Investors”).
WHEREAS, the Company and the Investors are executing and delivering this Agreement in reliance upon the exemption from securities registration afforded by Section 4(a)(2) of the Securities Act;
WHEREAS, the Company desires to sell to the Investors, and each Investor desires to purchase from the Company, severally and not jointly, upon the terms and subject to the conditions stated in this Agreement, shares (the “Shares”) of the Company’s common stock, par value $0.0001 per share (the “Common Stock”); and
WHEREAS, contemporaneously with the sale of the Shares, the parties hereto will execute and deliver a Registration Rights Agreement, substantially in the form attached hereto as Exhibit B, pursuant to which the Company will agree to provide certain registration rights in respect of the Shares under the Securities Act and applicable state securities laws.
NOW THEREFORE, in consideration of the mutual agreements, representations, warranties and covenants herein contained, the Company and each Investor, severally and not jointly, agree as follows:
1. Definitions. As used in this Agreement, the following terms shall have the following respective meanings:
“Affiliate” means, with respect to any Person, any other Person that, directly or indirectly through one or more intermediaries, controls, is controlled by or is under common control with such Person.
“Agreement” has the meaning set forth in the recitals.
“Amended and Restated Bylaws” means the Bylaws of the Company, as currently in effect.
“Amended and Restated Certificate of Incorporation” means the Restated Certificate of Incorporation of the Company, as currently in effect.
“Benefit Plan” or “Benefit Plans” means employee benefit plans as defined in Section 3(3) of ERISA and all other employee benefit practices or arrangements, including, without limitation, any such practices or arrangements providing severance pay, sick leave, vacation pay, salary continuation for disability, retirement benefits, deferred compensation, bonus pay, incentive pay, stock options or other stock-based compensation, hospitalization insurance,
medical insurance, life insurance, scholarships or tuition reimbursements, maintained by the Company or to which the Company is obligated to contribute for employees or former employees of the Company.
“Board of Directors” means the board of directors of the Company.
“Business Day” means any day except any Saturday, any Sunday, any day which is a federal legal holiday in the United States or any day on which banking institutions in the State of New York are authorized or required by law or other governmental action to close.
“Closing” has the meaning set forth in Section 2.1.
“Closing Date” has the meaning set forth in Section 2.1.
“Code” means the U.S. Internal Revenue Code of 1986, as amended.
“Common Stock” has the meaning set forth in the recitals.
“Common Stock Equivalents” means any securities of the Company that would entitle the holder thereof to acquire at any time Common Stock, including, without limitation, any debt, preferred stock, rights, options, warrants or other instrument that is at any time convertible into or exchangeable for, or otherwise entitles the holder thereof to receive, Common Stock.
“Company” has the meaning set forth in the recitals.
“Confidential Data” has the meaning set forth in Section 3.30.
“Disclosure Document” has the meaning set forth in Section 5.3.
“Drug Regulatory Agency” means the U.S. Food and Drug Administration (“FDA”) or other foreign, state, local or comparable governmental authority responsible for regulation of the research, development, testing, manufacturing, processing, storage, labeling, sale, marketing, advertising, distribution and importation or exportation of drug or biological products and drug or biological product candidates.
“Environmental Laws” has the meaning set forth in Section 3.15.
“ERISA” means the U.S. Employee Retirement Income Security Act of 1974, as amended.
“Exchange Act” means the U.S. Securities Exchange Act of 1934, as amended, and all of the rules and regulations promulgated thereunder.
“Financial Statements” has the meaning set forth in Section 3.8(b).
“Fundamental Representations” means the representations and warranties made by the Company in Sections 3.1 (Organization and Power), 3.2 (Capitalization), 3.4 (Authorization), 3.5
2
(Valid Issuance), 3.6 (No Conflict), 3.7 (Consents), 3.8 (SEC Filings; Financial Statements), 3.18 (Nasdaq Stock Market), 3.19 (Sarbanes-Oxley Act), 3.23 (Price Stabilization of Common Stock), 3.24 (Investment Company Act), 3.25 (General Solicitation; No Integration or Aggregation), 3.26 (Brokers and Finders), 3.27 (Reliance by the Investors) and 3.28 (No Additional Agreements).
“GAAP” has the meaning set forth in Section 3.8(b).
“GDPR” has the meaning set forth in Section 3.31.
“Governmental Authorizations” has the meaning set forth in Section 3.11.
“Health Care Laws” has the meaning set forth in Section 3.21.
“HIPAA” has the meaning set forth in Section 3.30.
“Indemnified Person” has the meaning set forth in Section 5.9.
“Intellectual Property” has the meaning set forth in Section 3.12.
“Investor” and “Investors” have the meanings set forth in the recitals.
“IT Systems” has the meaning set forth in Section 3.30.
“Material Adverse Effect” means any change, event, circumstance, development, condition, occurrence or effect that, individually or in the aggregate, (a) was, is, or would reasonably be expected to be, materially adverse to the business, financial condition, properties, assets, liabilities, stockholders’ equity or results of operations of the Company, or (b) materially delays or materially impairs the ability of the Company to comply, or prevents the Company from complying, with its obligations under this Agreement, the other Transaction Agreements, or with respect to the Closing, or would reasonably be expected to do so.
“Nasdaq” means the Nasdaq Stock Market LLC.
“National Exchange” means any of the following markets or exchanges on which the Common Stock is listed or quoted for trading on the date in question, together with any successor thereto: the NYSE American, The New York Stock Exchange, the Nasdaq Global Market, the Nasdaq Global Select Market and the Nasdaq Capital Market.
“Person” means an individual, partnership, corporation, limited liability company, business trust, joint stock company, trust, unincorporated association, joint venture or any other entity or organization.
“Personal Data” has the meaning set forth in Section 3.30.
“Privacy Laws” has the meaning set forth in Section 3.31.
3
“Privacy Statements” has the meaning set forth in Section 3.31.
“Process” or “Processing” has the meaning set forth in Section 3.31.
“Registration Rights Agreement” has the meaning set forth in Section 6.1(j).
“Regulatory Agencies” has the meaning set forth in Section 3.20.
“Rule 144” means Rule 144 promulgated by the SEC pursuant to the Securities Act, as such Rule may be amended from time to time, or any similar rule or regulation hereafter adopted by the SEC having substantially the same effect as such Rule.
“SEC” means the U.S. Securities and Exchange Commission.
“SEC Reports” means (a) the Company’s most recently filed Annual Report on Form 10-K and (b) all Quarterly Reports on Form 10-Q or Current Reports on Form 8-K filed or furnished (as applicable) by the Company following the end of the most recent fiscal year for which an Annual Report on Form 10-K has been filed and prior to the execution of this Agreement, together in each case with any documents incorporated by reference therein or exhibits thereto.
“Securities Act” means the U.S. Securities Act of 1933, as amended, and all of the rules and regulations promulgated thereunder.
“Shares” has the meaning set forth in the recitals.
“Short Sales” include, without limitation, (a) all “short sales” as defined in Rule 200 promulgated under Regulation SHO under the Exchange Act, whether or not against the box, and all types of direct and indirect stock pledges, forward sale contracts, options, puts, calls, short sales, swaps, “put equivalent positions” (as defined in Rule 16a-1(h) under the Exchange Act) and similar arrangements (including on a total return basis), and (b) sales and other transactions through non-U.S. broker dealers or non-U.S. regulated brokers (but shall not be deemed to include the location and/or reservation of borrowable shares of Common Stock).
“Studies” has the meaning set forth in Section 3.20.
“Tax” or “Taxes” means any and all federal, state, local, foreign and other taxes, levies, fees, imposts, duties and charges of whatever kind (including any interest, penalties or additions to the tax imposed in connection therewith or with respect thereto), whether or not imposed on the Company, including, without limitation, taxes imposed on, or measured by, income, franchise, profits or gross receipts, and also ad valorem, value added, sales, use, service, real or personal property, capital stock, license, payroll, withholding, employment, social security, workers’ compensation, unemployment compensation, utility, severance, production, excise, stamp, occupation, premium, windfall profits, transfer and gains taxes and customs duties.
“Tax Returns” means returns, reports, information statements and other documentation (including any additional or supporting material) filed or maintained, or required to be filed or
4
maintained, in connection with the calculation, determination, assessment or collection of any Tax and shall include any amended returns required as a result of examination adjustments made by the Internal Revenue Service or other Tax authority.
“Transaction Agreements” means this Agreement and the Registration Rights Agreement.
“Transfer Agent” means, with respect to the Common Stock, Equiniti Trust Company, LLC or such other financial institution that provides transfer agent services as the Company may engage from time to time.
2. Purchase and Sale of Shares.
2.1 Purchase and Sale. On the Closing Date, upon the terms and subject to the conditions set forth herein, the Company agrees to sell, and the Investors, severally and not jointly, agree to purchase, the number and type of Shares, for the aggregate purchase price, set forth opposite the Investor’s name on Exhibit A. The price per Share is $3.30.
2.2 Closing. Subject to the satisfaction or waiver of the conditions set forth in Section 5.6 of this Agreement, the closing of the purchase and sale of the Shares (the “Closing” and the date on which the Closing occurs, the “Closing Date”) shall occur remotely via the exchange of documents and signatures at such time as agreed to by the Company and the Investors but (i) in no event earlier than the first Business Day after the date of this Agreement and (ii) in no event later than the fifth Business Day after the date of this Agreement. At the Closing, the Shares shall be issued and registered in the name of the Investor, or in such nominee name(s) as designated by such Investor, representing the number of Shares to be purchased by the Investor at such Closing as set forth in Exhibit A against payment to the Company of the purchase price therefor in full, by wire transfer to the Company of immediately available funds, at or prior to the Closing, in accordance with wire instructions provided by the Company to the Investors no less than one Business Day prior to the Closing. On the Closing Date, the Company will cause the Transfer Agent to issue the Shares in book-entry form, free and clear of all restrictive and other legends (except as expressly provided in Section 4.10). In the event that the Closing has not occurred within one Business Day after the expected Closing Date, unless otherwise agreed by the Company and the Investors, the Company shall promptly (but no later than one Business Day thereafter) return the previously wired amounts to each respective Investor by wire transfer of United States dollars in immediately available funds to the account specified by each Investor, and any book entries for the Shares shall be deemed cancelled; provided that, unless this Agreement has been terminated pursuant to Section 7, such return of funds shall not terminate this Agreement or relieve such Investor of its obligation to purchase, or the Company of its obligation to issue and sell, the Shares at the Closing.
3. Representations and Warranties of the Company. Except as set forth in the SEC Reports (other than as to the Fundamental Representations, which are not so qualified), the Company hereby represents and warrants to each of the Investors that the statements contained in this Section 3 are true and correct as of the date of this Agreement and as of the Closing Date
5
(except for the representations and warranties that speak as of a specific date, which shall be made as of such date).
3.1 Organization and Power. The Company is a corporation duly organized, validly existing and in good standing under the laws of the State of Delaware, has the requisite power and authority to own, lease and operate its properties and to carry on its business as now conducted and described in the SEC Reports and is qualified to do business in each jurisdiction in which the character of its properties or the nature of its business requires such qualification, except where such failure to be in good standing or to have such power and authority or to so qualify would not reasonably be expected to have a Material Adverse Effect.
3.2 Capitalization. The Company’s disclosure of its authorized, issued and outstanding capital stock in the SEC Reports containing such disclosure was accurate in all material respects as of the date indicated in such SEC Reports. All of the issued and outstanding shares of Common Stock have been duly authorized and validly issued and are fully paid and non-assessable. None of the outstanding shares of capital stock of the Company were issued in violation of any preemptive or other similar rights of any securityholder of the Company which have not been waived, and such shares were issued in compliance in all material respects with applicable state and federal securities law and any rights of third parties.
3.3 Registration Rights. Except as set forth in the Transaction Agreements or as disclosed in the SEC Reports, the Company is presently not under any obligation, and has not granted any rights, to register under the Securities Act any of the Company’s presently outstanding securities or any of its securities that may hereafter be issued, other than such rights and obligations that have expired or been satisfied or waived.
3.4 Authorization. The Company has all requisite corporate power and authority to enter into the Transaction Agreements and to carry out and perform its obligations under the terms of the Transaction Agreements, including the issuance and sale of the Shares. All corporate action on the part of the Company, its officers, directors and stockholders necessary for the authorization of the Shares, the authorization, execution, delivery and performance of the Transaction Agreements and the consummation of the transactions contemplated herein, including the issuance and sale of the Shares, has been taken, including, without limitation, the approval of the Board of Directors (or a committee thereof) in accordance with Section 144(a)(1) of the Delaware General Corporation Law, as amended or superseded from time to time (the “DGCL”). This Agreement has been duly executed and delivered by the Company and, assuming the due authorization, execution and delivery by each Investor of this Agreement and that this Agreement constitutes the legal, valid and binding agreement of each Investor, this Agreement constitutes a legal, valid and binding obligation of the Company, enforceable against the Company in accordance with its terms, except as such enforceability may be limited by bankruptcy, insolvency, reorganization, moratorium and similar laws relating to or affecting creditors generally or by general equity principles (regardless of whether such enforceability is considered in a proceeding in equity or at law). Upon its execution by the Company and the other parties thereto and assuming that it constitutes legal, valid and binding agreements of the other parties thereto, the Registration Rights Agreement will constitute a legal,
6
valid and binding obligation of the Company, enforceable against the Company in accordance with its terms, except as such enforceability may be limited by bankruptcy, insolvency, reorganization, moratorium and similar laws relating to or affecting creditors generally or by general equity principles (regardless of whether such enforceability is considered in a proceeding in equity or at law).
3.5 Valid Issuance. The Shares being purchased by the Investors hereunder have been duly and validly authorized and, upon issuance pursuant to the terms of this Agreement against full payment therefor in accordance with the terms of this Agreement, will be duly and validly issued, fully paid and non-assessable and will be issued free and clear of any liens or other restrictions (other than those as provided in the Transaction Agreements or restrictions on transfer under applicable state and federal securities laws), and the holder of the Shares shall be entitled to all rights accorded to a holder of Common Stock. Subject to the accuracy of the representations and warranties made by the Investors in Section 4, the offer and sale of the Shares to the Investors is and will be in compliance with applicable exemptions from (i) the registration and prospectus delivery requirements of the Securities Act and (ii) the registration and qualification requirements of applicable securities laws of the states of the United States.
3.6 No Conflict. The execution, delivery and performance of the Transaction Agreements by the Company, the issuance and sale of the Shares and the consummation of the other transactions contemplated by the Transaction Agreements will not (i) violate any provision of the Amended and Restated Certificate of Incorporation or Amended and Restated Bylaws of the Company, (ii) conflict with or result in a violation of or default (with or without notice or lapse of time, or both) under, or give rise to a right of termination, cancellation or acceleration of any obligation, a change of control right or to a loss of a benefit under any agreement or instrument, credit facility, franchise, license, judgment, order, statute, law, ordinance, rule or regulations, applicable to the Company or its properties or assets, or (iii) result in a violation of any law, rule, regulation, order, judgment, injunction, decree or other restriction of any court or governmental authority to which the Company is subject (including federal and state securities laws and regulations) and the rules and regulations of any self-regulatory organization to which the Company or its securities are subject, or by which any property or asset of the Company is bound or affected, except, in the case of clauses (ii) and (iii), as would not, individually or in the aggregate, be reasonably expected to have a Material Adverse Effect.
3.7 Consents. Assuming the accuracy of the representations and warranties made by the Investors in Section 4, no consent, approval, authorization, filing with or order of or registration with, any court or governmental agency or body is required in connection with the authorization, execution or delivery by the Company of the Transaction Agreements, the issuance and sale of the Shares and the performance by the Company of its other obligations under the Transaction Agreements, except such as (a) have been or will be obtained or made under the Securities Act or the Exchange Act, (b) the filing of any requisite notices and/or application(s) to the National Exchange for the issuance and sale of the Shares and the listing of the Shares for trading or quotation, as the case may be, thereon in the time and manner required thereby, (c) customary post-closing filings with the SEC or pursuant to state securities laws in
7
connection with the offer and sale of the Shares by the Company in the manner contemplated herein, which will be filed on a timely basis, and (d) the filing of the registration statement required to be filed by the Registration Rights Agreement, or (e) such that the failure of which to obtain would not have a Material Adverse Effect. All notices, consents, authorizations, orders, filings and registrations which the Company is required to deliver or obtain prior to the Closing pursuant to the preceding sentence have been obtained or made or will be delivered or obtained or effected, and shall remain in full force and effect, on or prior to the Closing.
3.8 SEC Filings; Financial Statements.
(a) The Company has filed all forms, statements, certifications, reports and documents required to be filed by it with the SEC under Section 13, 14(a) and 15(d) of the Exchange Act for the one year preceding the date of this Agreement and is in compliance with General Instruction I.A.3 of Form S-3. As of the time it was filed with the SEC (or, if amended or superseded by a filing prior to the date of this Agreement, then on the date of such filing), each of the filed SEC Reports complied in all material respects with the applicable requirements of the Exchange Act, and, as of the time they were filed, none of the filed SEC Reports contained any untrue statement of a material fact or omitted to state a material fact required to be stated therein or necessary in order to make the statements therein, in light of the circumstances under which they were made, not misleading. There are no outstanding or unresolved comments from the SEC staff with respect to the SEC Reports. To the Company’s knowledge, none of the SEC Reports are the subject of an ongoing SEC review.
(b) The financial statements of the Company included in the SEC Reports (collectively, the “Financial Statements”) comply in all material respects with applicable accounting requirements and the rules and regulations of the SEC with respect thereto as in effect at the time of filing (or to the extent corrected by a subsequent restatement) and fairly present in all material respects the consolidated financial position of the Company as of the dates indicated, and the results of its operations and cash flows for the periods therein specified, all in accordance with United States generally accepted accounting principles (“GAAP”) (except as otherwise noted therein, and in the case of unaudited financial statements, as permitted by Form 10-Q of the SEC, and except that the unaudited financial statements may not contain footnotes and are subject to normal and recurring year-end adjustments) applied on a consistent basis throughout the periods therein specified (unless otherwise noted therein). Except as set forth in the Financial Statements filed prior to the date of this Agreement, the Company has not incurred any liabilities, contingent or otherwise, except (i) those incurred in the ordinary course of business, consistent with past practices since the date of such financial statements or (ii) liabilities not required under GAAP to be reflected in the Financial Statements, in either case, none of which, individually or in the aggregate, have had or would reasonably be expected to have a Material Adverse Effect.
3.9 Absence of Changes. Between December 31, 2024 and the date of this Agreement, (a) the Company has conducted its business only in the ordinary course of business and there have been no material transactions entered into by the Company (except for the execution and performance of this Agreement and the discussions, negotiations and transactions
8
related thereto); (b) no material change to any material contract or arrangement by which the Company is bound or to which any of its assets or properties is subject has been entered into that has not been disclosed in the SEC Reports; and (c) there has not been any other event or condition of any character that has had or would reasonably be expected to have a Material Adverse Effect; provided, however, that none of the following will be deemed in themselves, either alone or in combination, to constitute, and that none of the following will be taken into account in determining whether there has been or will be, a Material Adverse Effect under this Section 3.9:
(i) any change generally affecting the economy, financial markets or political, economic or regulatory conditions in the United States or any other geographic region in which the Company conducts business, provided that the Company is not disproportionately affected thereby;
(ii) general financial, credit or capital market conditions, including interest rates or exchange rates, or any changes therein, provided that the Company is not disproportionately affected thereby;
(iii) any change that generally affects industries in which the Company conducts business, provided that the Company is not disproportionately affected thereby;
(iv) earthquakes, hurricanes, tsunamis, tornadoes, floods, mudslides, fires or other natural disasters, weather conditions, global pandemics, including the COVID-19 pandemic and related strains, epidemic or similar health emergency, and other force majeure events in the United States or any other location, provided that the Company is not disproportionately affected thereby;
(v) national or international political or social conditions (or changes in such conditions), whether or not pursuant to the declaration of a national emergency or war, or the occurrence of any military or terrorist attack, provided that the Company is not disproportionately affected thereby;
(vi) material changes in laws after the date of this Agreement; and
(vii) in and of itself, any material failure by the Company to meet any published or internally prepared estimates of revenues, expenses, earnings or other economic performance for any period ending on or after the date of this Agreement (it being understood that the facts and circumstances giving rise to such failure may be deemed to constitute, and may be taken into account in determining whether there has been, a Material Adverse Effect to the extent that such facts and circumstances are not otherwise described in clauses (i)-(v) of this definition).
3.10 Absence of Litigation. There is no action, suit, proceeding, arbitration, claim, investigation, charge, complaint or inquiry pending or, to the Company’s knowledge, threatened against the Company that, individually or in the aggregate, has had or would reasonably be expected to have a Material Adverse Effect, nor are there any orders, writs, injunctions, judgments or decrees outstanding of any court or government agency or instrumentality and binding upon the Company that have had or would reasonably be expected to
9
have a Material Adverse Effect. The Company has not and, to the knowledge of the Company, no director or officer of the Company is, or within the last ten years has been, the subject of any action involving a claim of violation of or liability under federal or state securities laws relating to the Company or a claim of breach of fiduciary duty relating to the Company.
3.11 Compliance with Law; Permits. The Company is not in violation of, and has not received any notices of violations with respect to, any laws, statutes, ordinances, rules or regulations of any governmental body, court or government agency or instrumentality, except for violations which, individually or in the aggregate, have not had and would not reasonably be expected to have a Material Adverse Effect. The Company has all required licenses, permits, certificates and other authorizations (collectively, “Governmental Authorizations”) from such federal, state or local government or governmental agency, department or body that are currently necessary for the operation of the business of the Company as currently conducted, except where the failure to possess currently such Governmental Authorizations has not had and is not reasonably expected to have a Material Adverse Effect. The Company has not received any written (or, to the Company’s knowledge, oral) notice regarding any revocation or material modification of any such Governmental Authorization, which, individually or in the aggregate, if the subject of an unfavorable decision, ruling or finding, has or would reasonably be expected to result in a Material Adverse Effect.
3.12 Intellectual Property. The Company owns, or has rights to use, all material inventions, patent applications, patents, trademarks, trade names, service names, service marks, copyrights, trade secrets, know how (including unpatented and/or unpatentable proprietary of confidential information, systems or procedures) and other intellectual property as described in the SEC Reports necessary for, or used in the conduct of its business (including as described in the SEC Reports) (collectively, “Intellectual Property”), except where any failure to own, possess or acquire such Intellectual Property has not had, and would not, individually or in the aggregate, reasonably be expected to have a Material Adverse Effect. The Intellectual Property of the Company has not been adjudged by a court of competent jurisdiction to be invalid or unenforceable, in whole or in part. To the Company’s knowledge: (i) there are no third parties who have rights to any Intellectual Property, including no liens, security interests, or other encumbrances; and (ii) there is no infringement by third parties of any Intellectual Property, except, in each case, which, individually or in the aggregate, have not had and would not reasonably be expected to have a Material Adverse Effect. No action, suit, or other proceeding is pending, or, to the Company’s knowledge, is threatened: (A) challenging the Company’s rights in or to any Intellectual Property; (B) challenging the validity, enforceability or scope of any Intellectual Property; or (C) alleging that the Company infringes, misappropriates, or otherwise violates any patent, trademark, trade name, service name, copyright, trade secret or other proprietary rights of others, except, in each case, which, individually or in the aggregate, have not had and would not reasonably be expected to have a Material Adverse Effect. The Company has complied in all material respects with the terms of each agreement pursuant to which Intellectual Property has been licensed to the Company , and to the Company’s knowledge all such agreements are in full force and effect. To the Company’s knowledge, there are no material defects in any of the patents or patent applications included in the Intellectual Property. The
10
Company has taken all reasonable steps to protect, maintain and safeguard its Intellectual Property.
3.13 Employee Benefits. Except as would not be reasonably likely to result in a Material Adverse Effect, each Benefit Plan has been established and administered in accordance with its terms and in compliance with the applicable provisions of ERISA, the Code, the Patient Protection and Affordable Care Act of 2010, as amended, and other applicable laws, rules and regulations. The Company is in compliance with all applicable federal, state and local laws, rules and regulations regarding employment, except for any failures to comply that are not reasonably likely, individually or in the aggregate, to have a Material Adverse Effect. There is no labor dispute, strike or work stoppage against the Company pending or, to the knowledge of the Company, threatened which may interfere with the business activities of the Company, except where such dispute, strike or work stoppage is not reasonably likely, individually or in the aggregate, to have a Material Adverse Effect.
3.14 Taxes. The Company has filed all federal, state and foreign income Tax Returns and other Tax Returns required to have been filed under applicable law (or extensions have been duly obtained) and have paid all Taxes required to have been paid by them, except for those which are being contested in good faith and except where failure to file such Tax Returns or pay such Taxes would not, individually or in the aggregate, reasonably be expected to have a Material Adverse Effect. No assessment in connection with United States federal tax returns has been made against the Company. The charges, accruals and reserves on the books of the Company in respect of any income and corporation tax liability for any years not finally determined are adequate to meet any assessments or reassessments for additional income tax for any years not finally determined, except to the extent of any inadequacy that would not result in a Material Adverse Effect. No audits, examinations, or other proceedings with respect to any material amounts of Taxes of the Company are presently in progress or have been asserted or proposed in writing without subsequently being paid, settled or withdrawn. There are no liens on any of the assets of the Company. At all times since inception, the Company has been and continues to be classified as a corporation for U.S. federal income tax purposes. The Company has not been a United States real property holding corporation within the meaning of Code Section 897(c)-2 during the period specified in Code Section 897(c)(1)(A)(ii).
3.15 Environmental Laws. The Company (i) is in compliance with any and all applicable foreign, federal, state and local laws and regulations relating to the protection of human health and safety, the environment or hazardous or toxic substances or wastes, pollutants or contaminants (“Environmental Laws”), (ii) has received all permits and other Governmental Authorizations required under applicable Environmental Laws to conduct its business and (iii) is in compliance with all terms and conditions of any such permit, license or approval, except where such noncompliance with Environmental Laws, failure to receive required permits, licenses or other approvals or failure to comply with the terms and conditions of such permits, licenses or approvals would not, individually or in the aggregate, reasonably be expected to have a Material Adverse Effect. The Company has not received, since January 1, 2024, any written notice or other communication (in writing or otherwise), whether from a governmental authority or other Person, that alleges that the Company is not in compliance with any Environmental Law
11
and, to the knowledge of the Company, there are no circumstances that may prevent or interfere with the Company’s compliance in any material respects with any Environmental Law in the future, except where such failure to comply would not reasonably be expected to have a Material Adverse Effect. To the knowledge of the Company: (i) no current or (during the time a prior property was leased or controlled by the Company) prior property leased or controlled by the Company has received since January 1, 2024, any written notice or other communication relating to property owned or leased at any time by the Company, whether from a governmental authority, or other Person, that alleges that such current or prior owner or the Company is not in compliance with or violated any Environmental Law relating to such property and (ii) the Company has no material liability under any Environmental Law.
3.16 Title. The Company has good and marketable title to all personal property owned by it that is material to the business of the Company, free and clear of all liens, encumbrances and defects except such as do not materially affect the value of such property and do not interfere with the use made and proposed to be made of such property by the Company. Any real property and buildings held under lease by the Company are held under valid, subsisting and enforceable leases with such exceptions as are not material and do not interfere with the use made and proposed to be made of such property and buildings by the Company. The Company does not own any real property.
3.17 Insurance. The Company carries or is entitled to the benefits of insurance in such amounts and covering such risks that is customary for comparably situated companies and is adequate for the conduct of its business and the value of its real and personal properties (owned or leased) and tangible assets, and each of such insurance policies is in full force and effect and the Company is in compliance in all material respects with the terms of such insurance policies. Other than customary end-of-policy notifications from insurance carriers, since January 1, 2024, the Company has not received any notice or other communication regarding any actual or possible: (i) cancellation or invalidation of any material insurance policy or (ii) refusal or denial of any coverage, reservation of rights or rejection of any material claim under any insurance policy.
3.18 Nasdaq Stock Market. The issued and outstanding shares of Common Stock are registered pursuant to Section 12(b) of the Exchange Act and are listed for trading on the Nasdaq Global Select Market under the symbol “ABOS.” The Company is in compliance with all listing requirements of Nasdaq applicable to the Company. As of the date of this Agreement, there is no suit, action, proceeding or investigation pending or, to the knowledge of the Company, threatened against the Company by Nasdaq or the SEC, respectively, to prohibit or terminate the listing of the Common Stock on the Nasdaq Global Select Market or to deregister the Common Stock under the Exchange Act. The Company has taken no action as of the date of this Agreement that is designed to terminate the registration of the Common Stock under the Exchange Act.
3.19 Sarbanes-Oxley Act. The Company is, and since January 1, 2024 has been, in compliance in all material respects with all applicable requirements of the Sarbanes-Oxley Act of 2002 and applicable rules and regulations promulgated by the SEC thereunder.
12
3.20 Clinical Data and Regulatory Compliance. Except as would not reasonably be expected to result in a Material Adverse Effect: (i) the preclinical tests and clinical trials and other studies used to support regulatory approval (collectively, “Studies”) being conducted by the Company that are described in, or the results of which are referred to in, the SEC Reports were (and, if still pending, are being) conducted in all material respects in accordance with the protocols, procedures and controls designed and approved for such Studies and with standard medical and scientific research procedures; (ii) each description of the results of such Studies is accurate and complete in all material respects and fairly presents the data derived from such Studies, and the Company has no knowledge of any other studies the results of which are inconsistent with, or otherwise call into question, the results described or referred to in the SEC Reports; (iii) the Company has made all such filings and obtained all such approvals as may be required by the FDA or from any other U.S. federal, state or local government or foreign government or Drug Regulatory Agency, or Institutional Review Board, each having jurisdiction over biopharmaceutical products (collectively, the “Regulatory Agencies”) for the conduct of its business as described in the SEC Reports; (iv) the Company has not received any notice of, or correspondence from, any of the Regulatory Agencies requiring the termination or suspension of or imposing any clinical hold on any clinical trials that are described or referred to in the SEC Reports; and (v) the Company has operated and currently is in compliance in all material respects with all applicable rules, regulations and policies of the Regulatory Agencies.
3.21 Compliance with Health Care Laws. The Company is in compliance in all material respects with all Health Care Laws to the extent applicable to the Company’s current business and activities. For purposes of this Agreement, “Health Care Laws” means: (i) the Federal Food, Drug, and Cosmetic Act (21 U.S.C. Section 301 et seq.), and the regulations promulgated thereunder; (ii) all applicable federal, state, local and foreign health care fraud and abuse laws, including, without limitation, the Anti-Kickback Statute (42 U.S.C. Section 1320a-7b(b)); (iii) HIPAA, as amended by the Health Information Technology for Economic and Clinical Health Act (42 U.S.C. Section 17921 et seq.); (iv) the Patient Protection and Affordable Care Act of 2010, as amended by the Health Care and Education Reconciliation Act of 2010; (v) the European Union (“EU”) Clinical Trials Regulation (Regulation (EU) No. 536/2014); (vi) the EU Regulation regarding community procedures for authorization and supervision of medicinal products for human and veterinary use and establishing a European Medicines Agency (Regulation (EC) No. 726/2004); (vii) licensure, quality, safety and accreditation requirements under applicable federal, state, local or foreign laws or regulatory bodies; (viii) all other local, state, federal, national, supranational and foreign laws, relating to the regulation of the Company, and (ix) the regulations promulgated pursuant to such statutes and any state or non-U.S. counterpart thereof. The Company has not received written or, to the Company’s knowledge, oral notice of any claim, action, suit, proceeding, hearing, enforcement, investigation, arbitration or other action from any court or arbitrator or governmental or regulatory authority or third party alleging that any product operation or activity is in material violation of any Health Care Laws nor, to the Company’s knowledge, is any such claim, action, suit, proceeding, hearing, enforcement, investigation, arbitration or other action threatened. The Company has filed, maintained or submitted all material reports, documents, forms, notices, applications, records, claims, submissions and supplements or amendments as required by any Health Care Laws, and all such reports, documents, forms, notices, applications, records, claims, submissions and
13
supplements or amendments were complete and accurate on the date filed in all material respects (or were corrected or supplemented by a subsequent submission). The Company is not a party to any corporate integrity agreements, monitoring agreements, consent decrees, settlement orders, or similar agreements with or imposed by any governmental or regulatory authority. Additionally, neither the Company nor any of its employees, officers, directors, or, to the knowledge of the Company, agents has been excluded, suspended or debarred from participation in any U.S. federal health care program or human clinical research or, to the knowledge of the Company, is subject to a governmental inquiry, investigation, proceeding, or other similar action that would reasonably be expected to result in debarment, suspension, or exclusion.
3.22 Accounting Controls and Disclosure Controls and Procedures. The Company maintains a system of internal control over financial reporting (as defined in Rules 13a-15(f) and 15d-15(f) of the Exchange Act) that is designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with GAAP, including policies and procedures sufficient to provide reasonable assurance (i) that the Company maintains records that in reasonable detail accurately and fairly reflect the Company’s transactions and dispositions of assets, (ii) that transactions are recorded as necessary to permit preparation of financial statements in accordance with GAAP, (iii) that receipts and expenditures are made only in accordance with authorizations of management and the Board of Directors and (iv) regarding prevention or timely detection of the unauthorized acquisition, use or disposition of the Company’s assets that could have a material effect on the Company’s financial statements. Except as disclosed in the Company’s SEC Reports filed prior to the date of this Agreement, the Company has not identified any material weaknesses in the design or operation of the Company’s internal control over financial reporting. The Company’s “disclosure controls and procedures” (as defined in Rules 13a-15(e) and 15d-15(e) of the Exchange Act) are designed to provide reasonable assurance that all information (both financial and non-financial) required to be disclosed by the Company in the reports that it files or submits under the Exchange Act is recorded, processed, summarized and reported within the time periods specified in the rules and forms of the SEC, and that all such information is accumulated and communicated to the Company’s management as appropriate to allow timely decisions regarding required disclosure.
3.23 Price Stabilization of Common Stock. The Company has not taken, nor will it take, directly or indirectly, any action designed to stabilize or manipulate the price of the Common Stock to facilitate the sale or resale of the Shares.
3.24 Investment Company Act. The Company is not, and immediately after receipt of payment for the Shares will not be, an “investment company” within the meaning of the U.S. Investment Company Act of 1940, as amended.
3.25 General Solicitation; No Integration or Aggregation. Neither the Company nor any other person or entity authorized by the Company to act on its behalf has engaged in a general solicitation or general advertising (within the meaning of Regulation D of the Securities Act) of investors with respect to offers or sales of the Shares pursuant to this Agreement. The Company has not, directly or indirectly, sold, offered for sale, solicited offers to
14
buy or otherwise negotiated in respect of, any security (as defined in the Securities Act) which, to its knowledge, is or will be (i) integrated with the Shares sold pursuant to this Agreement for purposes of the Securities Act or (ii) aggregated with prior offerings by the Company for the purposes of the rules and regulations of the Nasdaq Global Select Market. Assuming the accuracy of the representations and warranties of the Investors set forth in Section 4, neither the Company nor any of its Affiliates or any Person acting on their behalf has, directly or indirectly, made any offers or sales of any Company security or solicited any offers to buy any Company security, under circumstances that would adversely affect reliance by the Company on Section 4(a)(2) for the exemption from registration for the transactions contemplated hereby.
3.26 Brokers and Finders. Neither the Company nor any other Person authorized by the Company to act on its behalf has retained, utilized or been represented by any broker or finder in connection with the transactions contemplated by this Agreement.
3.27 Reliance by the Investors. The Company has a reasonable basis for making each of the representations set forth in this Section 3. The Company acknowledges that each of the Investors will rely upon the truth and accuracy of, and the Company’s compliance with, the representations, warranties, agreements, acknowledgements and understandings of the Company set forth herein.
3.28 No Additional Agreements. There are no agreements or understandings between the Company and any Investor with respect to the transactions contemplated by the Transaction Agreements other than (i) as specified in the Transaction Agreements and (ii) any side letter agreements with any of the Investors, which side letters the Company has shared with all Investors.
3.29 Anti-Bribery and Anti-Money Laundering Laws. The Company and, to the knowledge of the Company, its officers, directors, supervisors, managers, agents, or employees are and have at all times been in compliance with, and its participation in the offering will not violate: (A) anti-bribery laws, including but not limited to, any applicable law, rule, or regulation of any locality, including but not limited to any law, rule, or regulation promulgated to implement the OECD Convention on Combating Bribery of Foreign Public Officials in International Business Transactions, signed December 17, 1997, including the U.S. Foreign Corrupt Practices Act of 1977, as amended, the U.K. Bribery Act 2010, or any other law, rule or regulation of similar purposes and scope or (B) anti-money laundering laws, including, but not limited to, applicable federal, state, international, foreign or other laws, regulations or government guidance regarding anti-money laundering, including, without limitation, Title 18 US. Code sections 1956 and 1957, the Patriot Act, the Bank Secrecy Act, and international anti-money laundering principles or procedures by an intergovernmental group or organization, such as the Financial Action Task Force on Money Laundering, of which the United States is a member and with which designation the United States representative to the group or organization continues to concur, all as amended, and any executive order, directive, or regulation pursuant to the authority of any of the foregoing, or any orders or licenses issued thereunder.
3.30 OISP. The Company is not and does not intend to become a “covered foreign person” within the meaning of the Outbound Investment Security Program. “Outbound
15
Investment Security Program” means the regulations implemented by the U.S. Department of the Treasury under Executive Order 14105 “Addressing United States Investments in Certain National Security Technologies and Products in Countries of Concern,” as codified at 31 C.F.R. Part 850.
3.31 Cybersecurity. The Company’s information technology assets and equipment, computers, systems, networks, hardware, software, websites, applications, and databases (collectively, “IT Systems”) are adequate for, and operate and perform in all material respects as required in connection with the operation of the business of the Company as currently conducted, and, to the knowledge of the Company, are free and clear of all material Trojan horses, time bombs, malware and other malicious code. The Company has implemented and maintained commercially reasonable physical, technical and administrative controls designed to maintain and protect the confidentiality, integrity, availability, privacy and security of all sensitive, confidential or regulated data (“Confidential Data”) used or maintained in connection with its businesses and Personal Data (defined below), and the integrity, availability continuous operation, redundancy and security of all IT Systems. “Personal Data” means the following data used in connection with the Company’s businesses and in its possession or control: (i) a natural person’s name, street address, telephone number, e-mail address, photograph, social security number or other tax identification number, driver’s license number, passport number, credit card number or bank information; (ii) information that identifies or may reasonably be used to identify an individual; (iii) any information that would qualify as “protected health information” under the Health Insurance Portability and Accountability Act of 1996, as amended by the Health Information Technology for Economic and Clinical Health Act (collectively, “HIPAA”); and (iv) any information that would qualify as “personal data,” “personal information” (or similar term) under the Privacy Laws. To the Company’s knowledge, there have been no breaches, outages or unauthorized uses of or accesses to the Company’s IT Systems, Confidential Data, or Personal Data that would require notification under Privacy Laws (as defined below).
3.32 Compliance with Data Privacy Laws. The Company is, and at all prior times has been, in material compliance with all applicable state, federal and foreign data privacy and security laws and regulations regarding the collection, use, storage, retention, disclosure, transfer, disposal, or any other processing (collectively “Process” or “Processing”) of Personal Data, including without limitation HIPAA, the EU General Data Protection Regulation (“GDPR”) (Regulation (EU) No. 2016/679), all other local, state, federal, national, supranational and foreign laws relating to the regulation of the Company, and the regulations promulgated pursuant to such statutes and any state or non-U.S. counterpart thereof (collectively, the “Privacy Laws”). To ensure material compliance with the Privacy Laws, the Company has in place, complies with, and takes all appropriate steps necessary to ensure compliance in all material respects with its policies and procedures relating to data privacy and security, and the Processing of Personal Data and Confidential Data (the “Privacy Statements”). The Company has, except as would not reasonably be expected, individually or in the aggregate, to result in a Material Adverse Effect, at all times since inception provided accurate notice of its Privacy Statements then in effect to its customers, employees, third party vendors and representatives. None of such disclosures made or contained in any Privacy Statements have been materially inaccurate, misleading, incomplete, or in material violation of any Privacy Laws.
16
3.33 Transactions with Affiliates and Employees. No relationship, direct or indirect, exists between or among the Company, on the one hand, and the directors, officers, stockholders, customers or suppliers of the Company, on the other hand, that is required to be described in the SEC Reports that is not so described.
4. Representations and Warranties of Each Investor. Each Investor, severally for itself and not jointly with any other Investor, represents and warrants to the Company that the statements contained in this Section 4 are true and correct as of the date of this Agreement and the Closing Date:
4.1 Organization. The Investor is duly organized, validly existing and in good standing under the laws of the jurisdiction of its organization and has the requisite power and authority to own, lease and operate its properties and to carry on its business as now conducted.
4.2 Authorization. The Investor has all requisite corporate or similar power and authority to enter into this Agreement and the other Transaction Agreements to which it will be a party and to carry out and perform its obligations hereunder and thereunder. All corporate, member or partnership action on the part of such Investor or its stockholders, members or partners necessary for the authorization, execution, delivery and performance of this Agreement and the other Transaction Agreements to which it will be a party and the consummation of the other transactions contemplated in this Agreement has been taken. The execution, delivery and performance by such Investor of the Transaction Agreements to which such Investor is a party has been duly authorized and each has been duly executed. Assuming this Agreement constitutes the legal and binding agreement of the Company, this Agreement constitutes a legal, valid and binding obligation of such Investor, enforceable against such Investor in accordance with its respective terms, except as such enforceability may be limited or otherwise affected by bankruptcy, insolvency, fraudulent conveyance, reorganization, moratorium and/or similar laws relating to or affecting the rights of creditors generally or by general equity principles (regardless of whether such enforceability is considered in a proceeding in equity or at law).
4.3 No Conflicts. The execution, delivery and performance of the Transaction Agreements by the Investor, the purchase of the Shares in accordance with their terms and the consummation by the Investor of the other transactions contemplated hereby will not conflict with or result in any violation of, breach or default by such Investor (with or without notice or lapse of time, or both) under, conflict with, or give rise to a right of termination, cancellation or acceleration of any obligation, a change of control right or to a loss of a material benefit under (i) any provision of the organizational documents of the Investor, including, without limitation, its incorporation or formation papers, bylaws, indenture of trust or partnership or operating agreement, as may be applicable or (ii) any agreement or instrument, undertaking, credit facility, franchise, license, judgment, order, ruling, statute, law, ordinance, rule or regulations, applicable to such Investor or its respective properties or assets, except, in the case of clause (ii), as would not, individually or in the aggregate, be reasonably expected to materially delay or hinder the ability of the Investor to perform its obligations under the Transaction Agreements.
4.4 Residency. The Investor’s residence (if an individual) or offices in which its investment decision with respect to the Shares was made (if an entity) are located at the
17
address immediately below the Investor’s name on Exhibit A, except as otherwise communicated by the Investor to the Company.
4.5 Brokers and Finders. The Investor has not retained, utilized or been represented by any broker or finder in connection with the transactions contemplated by this Agreement whose fees the Company would be required to pay.
4.6 Investment Representations and Warranties. The Investor hereby represents and warrants that, it (i) as of the date of this Agreement is, if an entity, a “qualified institutional buyer” (as defined in Rule 144A under the Securities Act) or an institutional “accredited investor” as that term is defined in Rule 501(a) under Regulation D promulgated pursuant to the Securities Act; or (ii) if an individual, is an “accredited investor” as that term is defined in Rule 501(a) of Regulation D of the Securities Act and has such knowledge and experience in financial and business matters as to be able to protect its own interests in connection with an investment in the Shares. The Investor further represents and warrants that (x) it is capable of evaluating the merits and risk of such investment, and (y) that it has not been organized for the purpose of acquiring the Shares and is an “institutional account” as defined by FINRA Rule 4512(c). The Investor understands and agrees that the offering and sale of the Shares has not been registered under the Securities Act or any applicable state securities laws and is being made in reliance upon federal and state exemptions for transactions not involving a public offering which depend upon, among other things, the bona fide nature of the investment intent and the accuracy of the Investor’s representations as expressed herein.
4.7 Intent. The Investor is purchasing the Shares solely for the Investor’s own account and not for the account of others, and not with a view to the resale or distribution of any part thereof in violation of the Securities Act, and the Investor has no present intention of selling, granting any participation in, or otherwise distributing the same in violation of the Securities Act without prejudice, however, to the Investor’s right at all times to sell or otherwise dispose of all or any part of such Shares in compliance with applicable federal and state securities laws. Notwithstanding the foregoing, if the Investor is purchasing the Shares as a fiduciary or agent for one or more investor accounts, the Investor has full investment discretion with respect to each such account, and the full power and authority to make the acknowledgements, representations and agreements herein on behalf of each owner of each such account. The Investor has no present arrangement to sell the Shares to or through any person or entity. The Investor understands that the Shares must be held indefinitely unless such Shares are resold pursuant to a registration statement under the Securities Act or an exemption from registration is available. Nothing contained herein shall be deemed a representation or warranty by the Investor to hold the Shares for any period of time.
4.8 Investment Experience; Ability to Protect Its Own Interests and Bear Economic Risks. The Investor acknowledges that it can bear the economic risk and complete loss of its investment in the Shares and has knowledge and experience in finance, securities, taxation, investments and other business matters as to be capable of evaluating the merits and risks of investments of the kind described in this Agreement and contemplated hereby, and the Investor has had an opportunity to seek, and has sought, such accounting, legal, business and tax
18
advice as the Investor has considered necessary to make an informed investment decision. The Investor acknowledges that the Investor (i) is a sophisticated investor, experienced in investing in private placements of equity securities and capable of evaluating investment risks independently, both in general and with regard to all transactions and investment strategies involving a security or securities and (ii) has exercised independent judgment in evaluating its participation in the purchase of the Shares. The Investor acknowledges that the Investor is aware that there are substantial risks incident to the purchase and ownership of the Shares, including those set forth in the Company’s filings with the SEC. Alone, or together with any professional advisor(s), the Investor has adequately analyzed and fully considered the risks of an investment in the Shares and determined that the Shares are a suitable investment for the Investor. The Investor is, at this time and in the foreseeable future, able to afford the loss of the Investor’s entire investment in the Shares and the Investor acknowledges specifically that a possibility of total loss exists.
4.9 Independent Investment Decision. The Investor understands that nothing in the Transaction Agreements or any other materials presented by or on behalf of the Company to the Investor in connection with the purchase of the Shares constitutes legal, tax or investment advice. The Investor has consulted such legal, tax and investment advisors as it, in such Investor’s sole discretion, has deemed necessary or appropriate in connection with its purchase of the Shares.
4.10 Shares Not Registered; Legends. The Investor acknowledges and agrees that the Shares are being offered in a transaction not involving any public offering within the meaning of the Securities Act, and the Investor understands that the Shares have not been registered under the Securities Act, by reason of their issuance by the Company in a transaction exempt from the registration requirements of the Securities Act, and that the Shares must continue to be held and may not be offered, resold, transferred, pledged or otherwise disposed of by the Investor unless a subsequent disposition thereof is registered under the Securities Act or is exempt from such registration and in each case in accordance with any applicable securities laws of any state of the United States. The Investor understands that the exemptions from registration afforded by Rule 144 (the provisions of which are known to it) promulgated under the Securities Act depend on the satisfaction of various conditions including, but not limited to, the time and manner of sale, the holding period and on requirements relating to the Company which are outside of the Investor’s control and which the Company may not be able to satisfy, and that, if applicable, Rule 144 may afford the basis for sales only in limited amounts. The Investor acknowledges and agrees that it has been advised to consult legal counsel prior to making any offer, resale, transfer, pledge or disposition of any of the Shares. The Investor acknowledges that no federal or state agency has passed upon or endorsed the merits of the offering of the Shares or made any findings or determination as to the fairness of this investment.
The Investor understands that any certificates or book entry notations evidencing the Shares may bear one or more legends in substantially the following form and substance:
“THE SECURITIES REPRESENTED HEREBY HAVE NOT BEEN REGISTERED UNDER THE SECURITIES ACT OF 1933, AS AMENDED (THE “SECURITIES ACT”), OR THE SECURITIES LAWS OF ANY STATE OF THE UNITED STATES.
19
THE SECURITIES HAVE BEEN ACQUIRED FOR INVESTMENT AND MAY NOT BE SOLD, TRANSFERRED OR ASSIGNED UNLESS (I) SUCH SECURITIES HAVE BEEN REGISTERED FOR SALE PURSUANT TO THE SECURITIES ACT, (II) SUCH SECURITIES MAY BE SOLD PURSUANT TO RULE 144, (III) THE COMPANY HAS RECEIVED AN OPINION OF COUNSEL REASONABLY SATISFACTORY TO IT THAT SUCH TRANSFER MAY LAWFULLY BE MADE WITHOUT REGISTRATION UNDER THE SECURITIES ACT, OR (IV) THE SECURITIES ARE TRANSFERRED WITHOUT CONSIDERATION TO AN AFFILIATE OF SUCH HOLDER OR A CUSTODIAL NOMINEE (WHICH FOR THE AVOIDANCE OF DOUBT SHALL REQUIRE NEITHER CONSENT NOR THE DELIVERY OF AN OPINION).”
In addition, the Shares may contain a legend regarding affiliate status of the Investor, if applicable.
4.11 No General Solicitation. The Investor acknowledges and agrees that the Investor is purchasing the Shares directly from the Company. The Investor became aware of this offering of the Shares directly from the Company as a result of a pre-existing, substantive relationship with the Company, and/or its advisors (including, without limitation, attorneys, accountants, bankers, consultants and financial advisors), agents, control persons, representatives, Affiliates, directors, officers, managers, members, and/or employees, and/or the representatives of such persons. The Shares were offered to Investor solely by direct contact between Investor and the Company or its representatives. Investor did not become aware of this offering of the Shares, nor were the Shares offered to Investor, by any other means, and neither the Company nor its representatives acted as investment advisor, broker or dealer to Investor. The Investor is not purchasing the Shares as a result of any general or public solicitation or general advertising, or publicly disseminated advertisement, article, notice or other communication regarding the Shares published in any newspaper, magazine or similar media or broadcast over television, radio or the internet or presented at any seminar or any other general solicitation or general advertisement, including any of the methods described in Section 502(c) of Regulation D under the Securities Act.
4.12 Access to Information. The Investor acknowledges and agrees that the Investor and the Investor’s professional advisor(s), if any, have had the opportunity to ask such questions, receive such answers and obtain such information from the Company regarding the Company, its business and the terms and conditions of the offering of the Shares as the Investor and the Investor’s professional advisor(s), if any, have deemed necessary to make an investment decision with respect to the Shares and that the Investor has independently made its own analysis and decision to invest in the Company. Neither such inquiries nor any other due diligence investigation conducted by the Investor shall modify, limit or otherwise affect the Investor’s right to rely on the Company’s representations and warranties contained in this Agreement.
4.13 Certain Trading Activities. Other than consummating the transaction contemplated hereby, the Investor has not, nor has any Person acting on behalf of or pursuant to any understanding with the Investor, directly or indirectly executed any purchases or sales,
20
including Short Sales, of the securities of the Company during the period commencing as of the time that the Investor was first contacted by the Company or any other Person regarding the transaction contemplated hereby and ending immediately prior to the date of this Agreement. Notwithstanding the foregoing, in the case of an Investor that is a multi-managed investment vehicle whereby separate portfolio managers manage separate portions of such Investor’s assets and the portfolio managers have no direct knowledge of the investment decisions made by the portfolio managers managing other portions of such Investor’s assets, the representation set forth above shall only apply with respect to the portion of the assets managed by the portfolio manager that made the investment decision to purchase the Shares covered by this Agreement. Other than to other Persons party to this Agreement and to its advisors and agents who had a need to know such information, the Investor has maintained the confidentiality of all disclosures made to it in connection with this transaction (including the existence and terms of this transaction). Notwithstanding the foregoing, for avoidance of doubt, nothing contained herein shall constitute a representation or warranty, or preclude any actions, with respect to the identification of the availability of, or securing of, available shares to borrow in order to effect Short Sales or similar transactions in the future.
5. Covenants.
5.1 Further Assurances. Each party agrees to cooperate with each other and their respective officers, employees, attorneys, accountants and other agents, and, generally, do such other reasonable acts and things in good faith as may be necessary to effectuate the intents and purposes of this Agreement, subject to the terms and conditions of this Agreement and compliance with applicable law, including taking reasonable action to facilitate the filing of any document or the taking of reasonable action to assist the other parties hereto in complying with the terms of this Agreement. The Investor acknowledges that the Company will rely on the acknowledgments, understandings, agreements, representations and warranties contained in this Agreement. Prior to the Closing, the Investor agrees to promptly notify the Company if any of the acknowledgments, understandings, agreements, representations and warranties set forth in Section 4 of this Agreement are no longer accurate.
5.2 Listing. The Company shall use commercially reasonable efforts to maintain the listing and trading of its Common Stock on the Nasdaq Global Select Market and, in accordance therewith, will use reasonable best efforts to comply in all material respects with the Company’s reporting, filing and other obligations under the rules and regulations of Nasdaq.
5.3 Disclosure of Transactions.
(a) The Company shall, by 9:00 a.m., New York City time, on the first (1st) Business Day immediately following the date of this Agreement (or, if the Agreement is executed on a Business Day prior to 8:00 a.m., New York City Time on the date of this Agreement, by 9:00 a.m., New York City time, on the date of this Agreement) file with the SEC a Current Report on Form 8-K (including all exhibits thereto, the “Disclosure Document”) disclosing (i) all material terms of the transactions contemplated hereby and by the other Transaction Agreements and attaching this Agreement and the other Transaction Agreements as exhibits to such Disclosure Document, and (ii) all material non-public information concerning
21
the Company disclosed to the Investors. Following the filing of the Disclosure Document, no Investor shall be in possession of any material non-public information concerning the Company disclosed to the Investors by the Company or its representatives. The Company understands and confirms that the Investors will rely on the foregoing representation in effecting securities transactions. Notwithstanding anything in this Agreement to the contrary, the Company shall not publicly disclose the name of any Investor or any of its Affiliates or advisers, or include the name of any Investor or any of its Affiliates or advisers in any press release, public announcement or filing with the SEC (other than any registration statement contemplated by the Registration Rights Agreement) or any regulatory agency, without the prior written consent of the Investor, except (i) as required by the federal securities law in connection with (A) any registration statement contemplated by the Registration Rights Agreement and (B) the filing of final Transaction Agreements with the SEC or pursuant to other routine proceedings of regulatory authorities, or (ii) to the extent such disclosure is required by law, at the request of the staff of the SEC or regulatory agency or under the regulations of the Nasdaq Global Select Market.
5.4 Integration. The Company shall not, and shall use its commercially reasonable efforts to ensure that no Affiliate of the Company shall, sell, offer for sale or solicit offers to buy or otherwise negotiate in respect of any security (as defined in Section 2 of the Securities Act) that will be integrated with the offer or sale of the Shares in a manner that would require the registration under the Securities Act of the sale of the Shares to the Investors, or that will be integrated with the offer or sale of the Shares for purposes of the rules and regulations of any National Exchange such that it would require stockholder approval prior to the closing of such other transaction unless stockholder approval is obtained before the closing of such subsequent transaction.
5.5 Removal of Legends.
(a) In connection with any sale, assignment, transfer or other disposition of the Shares by an Investor pursuant to Rule 144 or pursuant to any other exemption under the Securities Act such that the purchaser acquires freely tradable shares and upon compliance by the Investor with the requirements of this Agreement, if requested by the Investor by notice to the Company, the Company shall request the Transfer Agent to remove any restrictive legends related to the book entry account holding such shares and make a new, unlegended entry for such book entry shares sold or disposed of without restrictive legends as soon as reasonably practicable following any such request therefor from the Investor, provided that the Company has timely received from the Investor customary representations and other documentation reasonably acceptable to the Company in connection therewith. The Company shall be responsible for the fees of its Transfer Agent and its legal counsel associated with such legend removal.
(b) Subject to receipt from the Investor by the Company and the Transfer Agent of customary representations and other documentation reasonably acceptable to the Company and the Transfer Agent in connection therewith, upon the earliest of such time as the Shares (i) have been sold under the Securities Act pursuant to an effective registration
22
statement; (ii) have been sold pursuant to Rule 144, or (iii) are eligible for resale under Rule 144(b)(1) without the requirement for the Company to be in compliance with the current public information requirements under Rule 144(c)(1) (or any successor provision), the Company shall, in accordance with the provisions of this Section 5.5(b) and as soon as reasonably practicable following any request therefor from an Investor accompanied by such customary and reasonably acceptable documentation referred to above, (A) deliver to the Transfer Agent irrevocable instructions that the Transfer Agent shall make a new, unlegended entry for such book entry shares, and (B) cause its counsel to deliver to the Transfer Agent one or more opinions to the effect that the removal of such legends in such circumstances may be effected under the Securities Act if required by the Transfer Agent to effect the removal of the legend in accordance with the provisions of this Agreement.
5.6 Withholding Taxes. Each Investor agrees to furnish the Company with any information, representations and forms as shall reasonably be requested by the Company from time to time to assist the Company in complying with any applicable tax law (including any withholding obligations).
5.7 [RESERVED.]
5.8 No Conflicting Agreements. The Company will not take any action, enter into any agreement or make any commitment that would conflict or interfere in any material respect with the Company’s obligations to the Investors under the Transaction Agreements.
5.9 Indemnification.
(a) The Company agrees to indemnify and hold harmless each Investor and its Affiliates, and their respective directors, officers, trustees, members, managers, employees, investment advisers and agents (collectively, the “Indemnified Persons”), from and against any and all losses, claims, damages, liabilities and expenses (including without limitation reasonable and documented attorney fees and disbursements and other documented out-of-pocket expenses reasonably incurred in connection with investigating, preparing or defending any action, claim or proceeding, pending or threatened and the costs of enforcement thereof) to which such Person may become subject as a result of any breach of representation, warranty, covenant or agreement made by or to be performed on the part of the Company under the Transaction Agreements, and will reimburse any such Person for all such amounts as they are incurred by such Person solely to the extent such amounts have been finally judicially determined not to have resulted from such Person’s fraud or willful misconduct.
(b) Any person entitled to indemnification hereunder shall (i) give prompt written notice to the indemnifying party of any claim with respect to which it seeks indemnification and (ii) permit such indemnifying party to assume the defense of such claim with counsel reasonably satisfactory to the indemnified party; provided that any person entitled to indemnification hereunder shall have the right to employ separate counsel and to participate in the defense of such claim, but the fees and expenses of such counsel shall be at the expense of such person unless (a) the indemnifying party has agreed in writing to pay such fees or expenses, (b) the indemnifying party shall have failed to assume the defense of such claim and employ
23
counsel reasonably satisfactory to such person or (c) in the reasonable judgment of any such person, based upon written advice of its counsel, a conflict of interest exists between such person and the indemnifying party with respect to such claims (in which case, if the person notifies the indemnifying party in writing that such person elects to employ separate counsel at the expense of the indemnifying party, the indemnifying party shall not have the right to assume the defense of such claim on behalf of such person); and provided, further, that the failure of any indemnified party to give written notice as provided herein shall not relieve the indemnifying party of its obligations hereunder, except to the extent that such failure to give notice shall materially adversely affect the indemnifying party in the defense of any such claim or litigation. It is understood that the indemnifying party shall not, in connection with any proceeding in the same jurisdiction, be liable for fees or expenses of more than one separate firm of attorneys at any time for all such indemnified parties. No indemnifying party will, except with the consent of the indemnified party, which consent shall not be unreasonably withheld, conditioned or delayed, consent to entry of any judgment or enter into any settlement unless such judgment or settlement (i) imposes no liability or obligation on, (ii) includes as an unconditional term thereof the giving of a complete, explicit and unconditional release from the party bringing such indemnified claims of all liability of the indemnified party in respect of such claim or litigation in favor of, and (iii) does not include any admission of fault, culpability, wrongdoing, or malfeasance by or on behalf of, the indemnified party. No indemnified party will, except with the consent of the indemnifying party, which consent shall not be unreasonably withheld, conditioned or delayed, consent to entry of any judgment or enter into any settlement.
5.10 Subsequent Equity Sales. From the date of this Agreement until the earlier of (a) sixty (60) days after the Closing Date and (b) the Business Day immediately following the effective date of the registration statement filed pursuant to the Registration Rights Agreement, the Company shall not (A) issue shares of Common Stock or Common Stock Equivalents, (B) effect a reverse stock split, recapitalization, share consolidation, reclassification or similar transaction affecting the outstanding Common Stock or (C) file with the SEC a registration statement under the Securities Act relating to any shares of Common Stock or Common Stock Equivalents, except pursuant to the terms of the Registration Rights Agreement or as set forth in the following sentence. Notwithstanding the foregoing, the provisions of this Section 5.10 shall not apply to (i) the issuance of the Shares hereunder, (ii) the issuance of Common Stock or Common Stock Equivalents upon the conversion, exercise or vesting of any securities of the Company outstanding on the date of this Agreement or outstanding pursuant to clause (iii) below, (iii) the issuance of any Common Stock or Common Stock Equivalents pursuant to any Company stock-based compensation plans or in accordance with Nasdaq Stock Market Rule 5635(c)(4), or (iv) the filing of a registration statement on Form S-8 under the Securities Act to register the offer and sale of securities on an equity incentive plan or employee stock purchase plan.
5.11 [RESERVED].
24
6. Conditions of Closing.
6.1 Conditions to the Obligation of the Investors. The several obligations of each Investor to consummate the transactions to be consummated at the Closing, and to purchase and pay for the Shares being purchased by it at the Closing pursuant to this Agreement, are subject to the satisfaction or waiver in writing of the following conditions precedent:
(a) Representations and Warranties. The representations and warranties of the Company contained herein shall be true and correct in all material respects, except for those representation and warranties qualified by materiality or Material Adverse Effect, which shall be true and correct in all respects, as of the date of this Agreement and as of the Closing Date, as though made on and as of such date, except to the extent any such representation or warranty expressly speaks as of an earlier date, in which case such representation or warranty shall be true and correct in all material respects as of such earlier date, except for those representations and warranties qualified by materiality or Material Adverse Effect, which shall be true and correct in all respects as of such earlier date.
(b) Performance. The Company shall have performed in all material respects the obligations and conditions herein required to be performed or observed by the Company on or prior to the Closing Date.
(c) No Injunction. The purchase of and payment for the Shares by each Investor shall not be prohibited or enjoined by any law or governmental or court order or regulation and no such prohibition shall have been threatened in writing.
(d) Consents. The Company shall have obtained any and all consents, permits, approvals, registrations and waivers necessary for the consummation of the purchase and sale of the Shares, all of which shall be in full force and effect.
(e) Transfer Agent. The Company shall have furnished all required materials to the Transfer Agent to reflect the issuance of the Shares at the Closing.
(f) Adverse Changes. Since the date of this Agreement, no event or series of events shall have occurred that has had or would reasonably be expected to have a Material Adverse Effect.
(g) Opinion of Company Counsel. The Company shall have delivered to the Investors the opinion of Ropes & Gray LLP, dated as of the Closing Date, in customary form and substance to be reasonably agreed upon with the Investors and addressing such legal matters as the Investors and the Company reasonably agree.
(h) Compliance Certificate. An authorized officer of the Company shall have delivered to the Investors at the Closing Date a certificate certifying that the conditions specified in Sections 6.1(a) (Representations and Warranties), 6.1(b) (Performance), 6.1(c) (No Injunction), 6.1(d) (Consents), 6.1(e) (Transfer Agent), 6.1(f) (Adverse Changes), 6.1(k) (Listing Requirements) and 6.1(l) (No Injunction) of this Agreement have been fulfilled.
25
(i) Secretary’s Certificate. The Secretary of the Company shall have delivered to the Investors at the Closing Date a certificate certifying (i) the Amended and Restated Certificate of Incorporation; (ii) the Amended and Restated Bylaws; and (iii) resolutions of the Company’s Board of Directors (or an authorized committee thereof) approving this Agreement, the other Transaction Agreements, the transactions contemplated by this Agreement and the issuance of the Shares.
(j) Registration Rights Agreement. The Company shall have executed and delivered the Registration Rights Agreement in the form attached hereto as Exhibit B (the “Registration Rights Agreement”) to the Investors.
(k) Listing Requirements. No stop order or suspension of trading shall have been imposed by Nasdaq, the SEC or any other governmental or regulatory body with respect to public trading in the Common Stock. The Common Stock shall be listed on a National Exchange and shall not have been suspended, as of the Closing Date, by the SEC or the National Exchange from trading thereon nor shall suspension by the SEC or the National Exchange have been threatened, as of the Closing Date, in writing by the SEC or the National Exchange; and the Company shall have filed with Nasdaq a Notification Form: Listing of Additional Shares for the listing of the Shares and Nasdaq shall have raised no objection to such notice and the transactions contemplated hereby.
(l) No Injunction. No judgment, writ, order, injunction, award or decree of or by any court, or judge, justice or magistrate, including any bankruptcy court or judge, or any order of or by any Governmental Entity, shall have been issued, and no action or proceeding shall have been instituted by any Governmental Entity, enjoining or preventing the consummation of the transactions contemplated hereby or in the other Transaction Agreements.
(m) Payment. The Company shall have received payment, by wire transfer of immediately available funds, in the full amount of the purchase price for the number of Shares being purchased by each Investor at the Closing as set forth in Exhibit A.
6.2 Conditions to the Obligation of the Company. The obligation of the Company to consummate the transactions to be consummated at the Closing, and to issue and sell to each Investor the Shares to be purchased by it at the Closing pursuant to this Agreement, is subject to the satisfaction or waiver in writing of the following conditions precedent:
(a) Representations and Warranties. The representations and warranties of each Investor in Section 4 hereto shall be true and correct on and as of the Closing Date, with the same force and effect as though made on and as of the Closing Date and consummation of the Closing shall constitute a reaffirmation by the Investor of each of the representations, warranties, covenants and agreements of the Investor contained in this Agreement as of the Closing Date.
(b) Performance. Each Investor shall have performed or complied with in all material respects all obligations and conditions herein required to be performed or observed by such Investor on or prior to the Closing Date.
26
(c) Injunction. The purchase of and payment for the Shares by each Investor shall not be prohibited or enjoined by any law or governmental or court order or regulation.
(d) Registration Rights Agreement. Each Investor shall have executed and delivered the Registration Rights Agreement to the Company in the form attached as Exhibit B.
(e) Payment. The Company shall have received payment, by wire transfer of immediately available funds, in the full amount of the purchase price for the number of Shares being purchased by each Investor at the Closing as set forth in Exhibit A.
7. Termination.
7.1 Termination. The obligations of the Company, on the one hand, and the Investors, on the other hand, to effect the Closing shall terminate as follows:
(i) Upon the mutual written consent of the Company and the Investors that agreed to purchase a majority of the Shares prior to the Closing;
(ii) By the Company if any of the conditions set forth in Section 6.2 shall have become incapable of fulfillment, and shall not have been waived by the Company;
(iii) By an Investor (with respect to itself only) if any of the conditions set forth in Section 6.1 shall have become incapable of fulfillment, and shall not have been waived by such Investor; or
(iv) By either the Company or an Investor (with respect to itself only) if the Closing has not occurred on or prior to the fifth Business Day following the date of this Agreement; provided, however, that, in the case of clauses (ii) and (iii) above, the party seeking to terminate its obligation to effect the Closing shall not then be in breach of any of its representations, warranties, covenants or agreements contained in the Transaction Agreements if such breach has resulted in the circumstances giving rise to such party’s seeking to terminate its obligation to effect the Closing.
7.2 Notice. In the event of termination by the Company or the Investor of its obligations to effect the Closing pursuant to Section 7.1, written notice thereof shall be given to the other Investors by the Company. Nothing in this Section 7 shall be deemed to release any party from any liability for any breach by such party of the other terms and provisions of the Transaction Agreements or to impair the right of any party to compel specific performance by any other party of its other obligations under the Transaction Agreements.
8. Miscellaneous Provisions.
8.1 Public Statements or Releases. Except as set forth in Section 5.3, neither the Company nor any Investor shall make any public announcement with respect to the existence or terms of this Agreement or the transactions provided for herein without the prior consent of
27
the other party (which consent shall not be unreasonably withheld). Notwithstanding the foregoing, and subject to compliance with Section 5.3, nothing in this Section 8.1 shall prevent any party from making any public announcement it considers necessary in order to satisfy its obligations under the law, including applicable securities laws, or under the rules of any national securities exchange or securities market, in which case the Company shall allow the Investors reasonable time to comment on such release or announcement in advance of such issuance, and the Company will consider in good faith any Investor comments. The Company shall not include the name of the Investor in any press release or public announcement (which, for the avoidance of doubt, shall not include any filing with the SEC) without the prior written consent of the Investors, except as otherwise required by law or the applicable rules or regulations of any securities exchange or securities market, in which case the Company shall allow the Investors, to the extent reasonably practicable in the circumstances, reasonable time to comment on such release or announcement in advance of such issuance. Notwithstanding anything to the contrary in this Section 8.1, Investor review shall not be required for Company disclosures that are substantially consistent with prior Company disclosures.
8.2 Notices. Any notices or other communications required or permitted to be given hereunder shall be in writing and shall be deemed to be given (a) when delivered if personally delivered to the party for whom it is intended, (b) when delivered, if sent by electronic mail during normal business hours of the recipient, and if not sent during normal business hours, then on the recipient’s next Business Day, (c) three (3) days after having been sent by certified or registered mail, return-receipt requested and postage prepaid, or (d) one (1) Business Day after deposit with a nationally recognized overnight courier, freight prepaid, specifying next business day delivery, with written verification of receipt:
(a) If to the Company, addressed as follows:
Acumen Pharmaceuticals, Inc.
1210-1220 Washington St., Suite 210
Newton, Massachusetts 02465
Attention: Daniel O’Connell
with a copy (which shall not constitute notice):
Ropes & Gray LLP
Prudential Tower, 800 Boylston Street
Boston, MA 02199-3600
Attention: Thomas Danielski
Email:
Prudential Tower, 800 Boylston Street
Boston, MA 02199-3600
Attention: Thomas Danielski
Email:
(b) If to any Investor, at its address or e-mail address set forth on Exhibit A, or such address as subsequently modified by written notice given in accordance with this Section 8.2.
28
Any Person may change the address to which notices and communications to it are to be addressed by notification as provided for herein.
8.3 Consent to Electronic Notice. Each Investor consents to the delivery of any stockholder notice pursuant to the DGCL, as amended or superseded from time to time, by electronic mail pursuant to Section 232 of the DGCL (or any successor thereto) at the e-mail address set forth below the Investor’s name on the signature page or Exhibit A, as updated from time to time by notice to the Company. To the extent that any notice given by means of electronic mail is returned or undeliverable for any reason, the foregoing consent shall be deemed to have been revoked until a new or corrected e-mail address has been provided, and such attempted electronic notice shall be ineffective and deemed to not have been given. Each party agrees to promptly notify the other parties of any change in its e-mail address, and that failure to do so shall not affect the foregoing.
8.4 Severability. If any part or provision of this Agreement is held unenforceable or in conflict with the applicable laws or regulations of any jurisdiction, the invalid or unenforceable part or provisions shall be replaced with a provision which accomplishes, to the extent possible, the original business purpose of such part or provision in a valid and enforceable manner, and the remainder of this Agreement shall remain binding upon the parties hereto.
8.5 Governing Law; Submission to Jurisdiction; Venue; Waiver of Trial by Jury.
(a) This Agreement shall be governed by, and construed in accordance with, the laws of the State of New York without regard to choice of laws or conflicts of laws provisions thereof that would require the application of the laws of any other jurisdiction, except to the extent that mandatory principles of Delaware law may apply.
(b) The Company and each of the Investors hereby irrevocably and unconditionally:
(i) submits for itself and its property in any legal action or proceeding relating solely to this Agreement or the transactions contemplated hereby, to the general jurisdiction of the any state court or United States Federal court sitting in the Borough of Manhattan, City of New York in the State of New York;
(ii) consents that any such action or proceeding may be brought in such courts, and waives any objection that it may now or hereafter have to the venue of any such action or proceeding in any such court or that such action or proceeding was brought in an inconvenient court and agrees not to plead or claim the same to the extent permitted by applicable law;
(iii) agrees that service of process in any such action or proceeding may be effected by mailing a copy thereof by registered or certified mail (or any substantially similar
29
form of mail), postage prepaid, to the party, as the case may be, at its address set forth in Section 8.2 or at such other address of which the other party shall have been notified pursuant thereto;
(iv) agrees that nothing herein shall affect the right to effect service of process in any other manner permitted by law or shall limit the right to sue in any other jurisdiction for recognition and enforcement of any judgment or if jurisdiction in the courts referenced in the foregoing clause (i) are not available despite the intentions of the parties hereto;
(v) agrees that final judgment in any such suit, action or proceeding brought in such a court may be enforced in the courts of any jurisdiction to which such party is subject by a suit upon such judgment, provided that service of process is effected upon such party in the manner specified herein or as otherwise permitted by law;
(vi) agrees that to the extent that such party has or hereafter may acquire any immunity from jurisdiction of any court or from any legal process with respect to itself or its property, such party hereby irrevocably waives such immunity in respect of its obligations under this Agreement, to the extent permitted by law; and
(vii) irrevocably and unconditionally waives trial by jury in any legal action or proceeding in relation to this Agreement.
8.6 Waiver. No waiver of any term, provision or condition of this Agreement, whether by conduct or otherwise, in any one or more instances, shall be deemed to be, or be construed as, a further or continuing waiver of any such term, provision or condition or as a waiver of any other term, provision or condition of this Agreement.
8.7 Expenses. Except as expressly set forth in the Transaction Agreements to the contrary, each party shall pay its own out-of-pocket fees and expenses, including the fees and expenses of attorneys, accountants and consultants employed by such party, incurred in connection with the proposed investment in the Shares and the consummation of the transactions contemplated thereby; provided, however, that the Company shall pay all Transfer Agent fees (including, without limitation, any fees required for same-day processing of any instruction letter delivered by the Company), stamp taxes and other taxes (other than income taxes) and duties levied in connection with the delivery of any Shares to the Investors.
8.8 Assignment. None of the parties may assign its rights or obligations under this Agreement or designate another person (i) to perform all or part of its obligations under this Agreement or (ii) to have all or part of its rights and benefits under this Agreement, in each case without the prior written consent of (x) the Company, in the case of an Investor, and (y) the Investors, in the case of the Company, provided that an Investor may, without the prior consent of the Company, assign its rights to purchase the Shares hereunder to any of its Affiliates or to any other investment funds or accounts managed or advised by the investment manager who acts on behalf of such Investor (provided each such assignee agrees to be bound by the terms of this Agreement and makes the same representations and warranties set forth in Section 4 ). In the event of any assignment in accordance with the terms of this Agreement, the assignee shall specifically assume and be bound by the provisions of this Agreement by executing a writing
30
agreeing to be bound by and subject to the provisions of this Agreement and shall deliver an executed counterpart signature page to this Agreement and, notwithstanding such assumption or agreement to be bound hereby by an assignee, no such assignment shall relieve any party assigning any interest hereunder from its obligations or liability pursuant to this Agreement.
8.9 Confidential Information.
(a) Each Investor covenants that until such time as the transactions contemplated by this Agreement and any material non-public information provided to such Investor are publicly disclosed by the Company, such Investor will maintain the confidentiality of all disclosures made to it in connection with this transaction (including the existence and terms of this transaction), other than to such Investor’s outside attorney, accountant, auditor or investment advisor only to the extent necessary to permit evaluation of the investment, and the performance of the necessary or required tax, accounting, financial, legal, or administrative tasks and services and other than as may be required by law.
(b) The Company may request from the Investors such reasonable and customary additional information as the Company may deem necessary to evaluate the eligibility of the Investor to acquire the Shares, and the Investor shall promptly provide such information as may reasonably be requested to the extent readily available; provided, that the Company agrees to keep any such information provided by the Investor confidential, except (i) as required by the federal securities laws, rules or regulations and (ii) to the extent such disclosure is required by other laws, rules or regulations, at the request of the staff of the SEC or regulatory agency or under the regulations of Nasdaq. The Investor acknowledges that the Company may file a copy of this Agreement and the Registration Rights Agreement with the SEC as exhibit to a periodic report or a registration statement of the Company.
8.10 [RESERVED].
8.11 Third Parties. Nothing in this Agreement, express or implied, is intended to confer on any Person other than the parties to this Agreement any rights, remedies, claims, benefits, obligations or liabilities under or by reason of this Agreement, and no Person that is not a party to this Agreement (including, without limitation, any partner, member, shareholder, director, officer, employee or other beneficial owner of any party to this Agreement, in its own capacity as such or in bringing a derivative action on behalf of a party to this Agreement) shall have any standing as a third party beneficiary with respect to this Agreement or the transactions contemplated hereby. Notwithstanding the foregoing, the Indemnified Persons are intended third-party beneficiaries of Section 5.9 of this Agreement.
8.12 Independent Nature of Investors’ Obligations and Right. The obligations of each Investor under this Agreement are several and not joint with the obligations of any other Investor, and no Investor shall be responsible in any way for the performance obligations of any other Investor under this Agreement. Nothing contained herein, and no action taken by any Investor pursuant hereto, shall be deemed to constitute the Investors as, and the Company acknowledges that the Investors do not so constitute, a partnership, an association, a joint venture or any other kind of entity, or create a presumption that the Investors are in any way acting in
31
concert or as a group, and the Company will not assert any such claim with respect to such obligations or the transactions contemplated by this Agreement. The Company acknowledges and each Investor confirms that it has independently participated in the negotiation of the transaction contemplated hereby with the advice of its own counsel and advisors. Each Investor also acknowledges that Ropes & Gray LLP has not rendered legal advice to such Investor. Each Investor shall be entitled to independently protect and enforce its rights, including, without limitation, the rights arising out of this Agreement, and it shall not be necessary for any other Investor to be joined as an additional party in any proceeding for such purpose. The Company has elected to provide all Investors with the same terms and Transaction Agreements for the convenience of the Company and not because it was required or requested to do so by any Investor.
8.13 Headings. The titles, subtitles and headings in this Agreement are for convenience of reference and shall not form part of, or affect the interpretation of, this Agreement.
8.14 Counterparts. This Agreement may be executed in two or more identical counterparts, all of which shall be considered one and the same agreement and shall become effective when counterparts have been signed by each party and delivered to the other party; provided that a facsimile or pdf signature including any electronic signatures complying with the U.S. federal ESIGN Act of 2000, e.g., www.docusign.com shall be considered due execution and shall be binding upon the signatory thereto with the same force and effect as if the signature were an original, not a facsimile or pdf (or other electronic reproduction of a) signature.
8.15 Entire Agreement; Amendments. This Agreement and the other Transaction Agreements (including all schedules and exhibits hereto and thereto), together with any side letter agreements with any of the Investors, constitute the entire agreement between the parties hereto respecting the subject matter of this Agreement and supersedes all prior agreements, negotiations, understandings, representations and statements respecting the subject matter of this Agreement, whether written or oral. No amendment, modification, alteration, or change in any of the terms of this Agreement shall be valid or binding upon the parties hereto unless made in writing and duly executed by the Company and the Investors of at least a majority in interest of the Shares then held by the Investors, provided that prior to the Closing the consent of all Investors shall be required. Notwithstanding the foregoing, this Agreement may not be amended and the observance of any term of this Agreement may not be waived with respect to any Investor without the written consent of such Investor unless such amendment or waiver applies to all Investors in the same fashion. The Company, on the one hand, and each Investor, on the other hand, may by an instrument signed in writing by such parties waive the performance, compliance or satisfaction by such Investor or the Company, respectively, with any term or provision of this Agreement or any condition hereto to be performed, complied with or satisfied by such Investor or the Company, respectively.
8.16 Survival. The covenants, representations and warranties made by each party hereto contained in this Agreement shall survive the Closing and the delivery of the Shares
32
in accordance with their respective terms. Each Investor shall be responsible only for its own representations, warranties, agreements and covenants hereunder.
8.17 Contract Interpretation. This Agreement is the joint product of each Investor and the Company and each provision of this Agreement has been subject to the mutual consultation, negotiation and agreement of such parties and shall not be construed for or against any party hereto.
8.18 Arm’s Length Negotiations. For the avoidance of doubt, the parties acknowledge and confirm that the terms and conditions of the Shares were determined as a result of arm’s-length negotiations.
[Remainder of Page Intentionally Left Blank.]
33
IN WITNESS WHEREOF, the parties hereto have executed this Agreement as of the day and year first above written.
COMPANY:
ACUMEN PHARMACEUTICALS, INC.
ACUMEN PHARMACEUTICALS, INC.
By: _______________________
Name: Daniel O’Connell
Title: Chief Executive Officer
Name: Daniel O’Connell
Title: Chief Executive Officer
[Signature Page to Securities Purchase Agreement] | ||
IN WITNESS WHEREOF, the parties hereto have executed this Agreement as of the day and year first above written.
INVESTOR:
By: ______________________
Name: ____________________
Title: _______________________
[Signature Page to Securities Purchase Agreement] | ||
EXHIBIT A
INVESTORS
INVESTORS
1
EXHIBIT B
REGISTRATION RIGHTS AGREEMENT
1
Exhibit 10.2
Execution Version
REGISTRATION RIGHTS AGREEMENT
THIS REGISTRATION RIGHTS AGREEMENT (this “Agreement”), dated as of March 13, 2026, is entered into by and among Acumen Pharmaceuticals, Inc., a Delaware corporation (the “Company”), and the several investors signatory hereto (individually as an “Investor” and collectively together with their respective permitted assigns, the “Investors”). Capitalized terms used herein and not otherwise defined herein shall have the respective meanings set forth in the Securities Purchase Agreement by and among the parties hereto, dated as of the date hereof (as amended, restated, supplemented or otherwise modified from time to time, the “Purchase Agreement”).
WHEREAS:
A. Upon the terms and subject to the conditions of the Purchase Agreement, the Company has agreed to issue to the Investors, and the Investors have agreed to purchase, severally and not jointly, an aggregate of up to $35,750,000 of shares (the “Shares”) of the Company’s common stock, par value $0.0001 per share (the “Common Stock”) pursuant to the Purchase Agreement.
B. To induce the Investors to enter into the Purchase Agreement, the Company has agreed to provide certain registration rights under the U.S. Securities Act of 1933, as amended, and the rules and regulations thereunder, or any similar successor statute (collectively, the “Securities Act”), and applicable state securities laws.
NOW, THEREFORE, in consideration of the promises and the mutual covenants contained herein and other good and valuable consideration, the receipt and sufficiency of which are hereby acknowledged, the Company and the Investors hereby agree as follows:
1. DEFINITIONS.
For purposes of this Agreement, the following terms shall have the following meanings:
(a) “Person” means an individual, partnership, corporation, limited liability company, business trust, joint stock company, trust, unincorporated association, joint venture or any other entity or organization.
(b) “Register,” “Registered,” and “Registration” refer to a registration effected by preparing and filing one or more registration statements of the Company in compliance with the Securities Act and providing for offering securities on a continuous basis, and the declaration or ordering of effectiveness of such registration statement(s) by the U.S. Securities and Exchange Commission (the “SEC”).
(c) “Registrable Securities” means the Shares and any Common Stock issued or issuable with respect to the Shares as a result of any stock split or subdivision, stock dividend, recapitalization, exchange or similar event. Registrable Securities shall cease to be Registrable Securities upon the date on which the Investors shall have resold all the Registrable Securities covered by the Registration Statement.
(d) “Registration Expenses” means all registration and filing fee expenses incurred by the Company in effecting any registration pursuant to this Agreement, including (i) all registration, qualification, and filing fees, printing expenses, and any other fees and expenses associated with filings
required to be made with the SEC, FINRA or any other regulatory authority, (ii) all fees and expenses in connection with compliance with or clearing the Registrable Securities for sale under any securities or “Blue Sky” laws, (iii) all printing, duplicating, word processing, messenger, telephone, facsimile and delivery expenses, and (iv) all fees and disbursements of counsel for the Company and of all independent certified public accountants of the Company (including the expenses of any special audit and cold comfort letters required by or incident to such performance).
(e) “Registration Statement” means any registration statement of the Company filed with, or to be filed with, the SEC under the Securities Act, that Registers the Registrable Securities, including the related prospectus, amendments and supplements to such registration statement, including any pre- and post-effective amendments, and all exhibits and all material incorporated by reference in such registration statement as may be necessary to comply with applicable securities laws. “Registration Statement” shall also include a New Registration Statement, as amended when each became effective, including all documents filed as part thereof or incorporated by reference therein, and including any information contained in a prospectus subsequently filed with the SEC.
(f) “Selling Expenses” means all underwriting discounts and selling commissions applicable to the sale of Registrable Securities and all similar fees and commissions relating to the Investors’ disposition of the Registrable Securities.
2. REGISTRATION.
(a) Mandatory Registration. The Company shall, as promptly as reasonably practicable and in any event no later than two Business Days after the date on which the Company files with the SEC its Annual Report on Form 10-K for the fiscal year ended December 31, 2025 (the “Filing Deadline”), prepare and file with the SEC an initial Registration Statement (the “Initial Registration Statement”) to Register the resale of all Registrable Securities. Before filing the Registration Statement, the Company shall furnish to the Investors a copy of the Registration Statement. The Investors and their counsel shall have at least three Business Days prior to the anticipated filing date of a Registration Statement to review and comment upon such Registration Statement and any amendment or supplement to such Registration Statement and any related prospectus, prior to its filing with the SEC. Subject to any SEC comments, such Registration Statement shall include the plan of distribution substantially in the form attached hereto as Exhibit A. The Company shall (a) use commercially reasonable efforts to address in each such document prior to being so filed with the SEC such comments as the Investor or its counsel reasonably proposed by the Investor, and (b) not file any Registration Statement or related prospectus or any amendment or supplement thereto containing information regarding the Investor to which Investor reasonably objects, unless such information is required to comply with any applicable law or regulation. The Investors shall furnish all information reasonably requested by the Company and as shall be reasonably required in connection with any registration referred to in this Agreement.
(b) Effectiveness. The Company shall use its reasonable best efforts to have the Initial Registration Statement and any amendment declared effective by the SEC at the earliest possible date, but in any event no later than the earlier of (i) the 75th calendar day following the initial filing date of the Initial Registration Statement if the SEC notifies the Company that it will “review” the Initial Registration Statement and (ii) the fifth Business Day after the date the Company is notified (orally or in writing, whichever is earlier) by the SEC that the Initial Registration Statement will not be “reviewed” or will not be subject to further review (the “Effectiveness Deadline”). The Company shall notify the Investor by e-mail as promptly as practicable, and in any event, within 24 hours, after the Registration
2
Statement is declared effective or is supplemented and shall provide the Investor with copies of any related prospectus to be used in connection with the sale or other disposition of the securities covered thereby. The Company shall use reasonable best efforts to keep the Initial Registration Statement continuously effective pursuant to Rule 415 promulgated under the Securities Act and available for the resale by the Investors of all of the Registrable Securities covered thereby at all times until the earliest to occur of the following events: (i) the date on which the Investors shall have resold all the Registrable Securities covered thereby; and (ii) the date on which the Registrable Securities may be resold by the Investors without registration and without regard to any volume or manner-of-sale limitations by reason of Rule 144, without the requirement for the Company to be in compliance with the current public information requirement under Rule 144 under the Securities Act or any other rule of similar effect (the “Registration Period”). The Initial Registration Statement (including any amendments or supplements thereto and prospectuses contained therein) shall not contain any untrue statement of a material fact or omit to state a material fact required to be stated therein, or necessary to make the statements therein, in light of the circumstances in which they were made, not misleading.
(c) Sufficient Number of Shares Registered. In the event the number of shares available under the Initial Registration Statement at any time is insufficient to cover the Registrable Securities, the Company shall, to the extent necessary and permissible, amend the Initial Registration Statement or file a new registration statement (together with any prospectuses or prospectus supplements thereunder, a “New Registration Statement”), so as to cover all of such Registrable Securities as soon as reasonably practicable, but in any event not later than ten Business Days after the necessity therefor arises (the “New Registration Filing Deadline”). The Company shall use its reasonable best efforts to have such amendment and/or New Registration Statement become effective as soon as reasonably practicable following the filing thereof but no later than the earlier of (i) the 75th calendar day following the initial filing date of the New Registration Statement if the SEC notifies the Company that it will “review” the New Registration Statement and (ii) the fifth Business Day after the date the Company is notified (orally or in writing, whichever is earlier) by the SEC that the New Registration Statement will not be “reviewed” or will not be subject to further review (the earlier of such dates, the “New Registration Effectiveness Deadline”). The provisions of Section 2(a) and (b) shall apply to the New Registration Statement, except as modified hereby.
(d) Liquidated Damages. If (i) the Initial Registration Statement has not been filed by the Filing Deadline, (ii) the Initial Registration Statement has not been declared effective by the Effectiveness Deadline, (iii) the New Registration Statement has not been filed by the New Registration Filing Deadline, (iv) the New Registration Statement has not been declared effective by the New Registration Effectiveness Deadline or (v) after any Registration Statement has been declared effective by the SEC, sales cannot be made pursuant to such Registration Statement for any reason (including without limitation by reason of a stop order, or the Company’s failure to update such Registration Statement), but excluding any Allowed Delay (as defined below) or, if the Registration Statement is on Form S-1, for a period of 20 days following the date on which the Company files a post-effective amendment to incorporate the Company’s Annual Report on Form 10-K (a “Maintenance Failure”), then the Company will make pro rata payments to each Investor then holding Registrable Securities, as liquidated damages and not as a penalty, in an amount equal to 1.0% of the aggregate amount paid pursuant to the Purchase Agreement by such Investor for such Registrable Securities then held by such Investor for each 30-day period or pro rata for any portion thereof during which the failure continues (the “Blackout Period”). Such payments shall constitute the Investors’ exclusive monetary remedy for such events, but shall not affect the right of the Investors to seek injunctive relief. The amounts payable as liquidated damages pursuant to this paragraph shall be paid in cash no later than five Business Days after each such 30-day period following the commencement of the Blackout Period until the termination of the Blackout Period
3
(the “Blackout Period Payment Date”). Interest shall accrue at the rate of 1.0% per month on any such liquidated damages payments that shall not be paid by the Blackout Period Payment Date until such amount is paid in full. Notwithstanding the above, in no event shall the aggregate amount of liquidated damages (or interest thereon) paid under this Agreement to any Investor exceed, in the aggregate, 5.0% of the aggregate purchase price of the Shares purchased by such Investor under the Purchase Agreement. Notwithstanding anything in this Section 2(d) to the contrary, during any periods that the Company is unable to meet its obligations hereunder with respect to the registration of the Registrable Securities because any Investor fails to furnish information required to be provided pursuant to Section 2(a) or Section 4(a) within three Business Days of the Company’s request, any liquidated damages that would otherwise accrue as to such Investor only shall be tolled until such information is delivered to the Company.
(e) Allowable Delays. On no more than two occasions and for not more than 30 consecutive days or for a total of not more than 60 days in any 12 month period, the Company may delay the effectiveness of the Initial Registration Statement or any other Registration Statement, or suspend the use of any prospectus included in any Registration Statement, in the event that the Company determines in good faith that such delay or suspension is necessary to (A) delay the disclosure of material non-public information concerning the Company, the disclosure of which at the time is not, in the good faith opinion of the Company, in the best interests of the Company or (B) amend or supplement the affected Registration Statement or the related prospectus so that such Registration Statement or prospectus shall not include an untrue statement of a material fact or omit to state a material fact required to be stated therein or necessary to make the statements therein, in the case of the prospectus in light of the circumstances under which they were made, not misleading (an “Allowed Delay”); provided, that the Company shall promptly (a) notify each Investor in writing of the commencement of an Allowed Delay, but shall not (without the prior written consent of an Investor) disclose to such Investor any material non-public information giving rise to an Allowed Delay, (b) advise the Investors in writing to cease all sales under the Registration Statement until the end of the Allowed Delay and (c) use commercially reasonable efforts to terminate an Allowed Delay as promptly as practicable.
(f) Rule 415; Cutback. If at any time the SEC takes the position that the offering of some or all of the Registrable Securities in any Registration Statement is not eligible to be made on a delayed or continuous basis under the provisions of Rule 415 under the Securities Act (provided, however, the Company shall be obligated to use reasonable best efforts to advocate with the SEC for the registration of all of the Registrable Securities) or requires any Investor to be named as an “underwriter,” the Company shall (i) promptly notify each holder of Registrable Securities thereof and (ii) make commercially reasonable efforts to persuade the SEC that the offering contemplated by such Registration Statement is a valid secondary offering and not an offering “by or on behalf of the issuer” as defined in Rule 415 and that none of the Investors is an “underwriter.” The Investors shall have the right to select one legal counsel, at such Investor’s expense, to review and oversee any registration or matters pursuant to this Section 2(f), including participation in any meetings or discussions with the SEC regarding the SEC’s position and to comment on any written submission made to the SEC with respect thereto. No such written submission with respect to this matter shall be made to the SEC to which any Investor’s counsel reasonably objects. In the event that, despite the Company’s reasonable best efforts and compliance with the terms of this Section 2(f), the SEC refuses to alter its position, the Company shall (i) remove from such Registration Statement such portion of the Registrable Securities (the “Cut Back Shares”) and/or (ii) agree to such restrictions and limitations on the registration and resale of the Registrable Securities as the SEC may require to assure the Company’s compliance with the requirements of Rule 415 (collectively, the “SEC Restrictions”); provided, however, that the Company shall not name any Investor as an “underwriter” in such Registration Statement without the prior written consent of such Investor
4
(provided that, in the event an Investor withholds such consent, the Company shall have no obligation hereunder to include any Registrable Securities of such Investor in any Registration Statement covering the resale thereof until such time as the SEC no longer requires such Investor to be named as an “underwriter” in such Registration Statement or such Investor otherwise consents in writing to being so named). Any cut-back imposed on the Investors pursuant to this Section 2(f) shall be allocated among the Investors on a pro rata basis and shall be applied first to any of the Registrable Securities of such Investor as such Investor shall designate, unless the SEC Restrictions otherwise require or provide or the Investors otherwise agree. No liquidated damages shall accrue as to any Cut Back Shares until such date as the Company is able to effect the registration of such Cut Back Shares in accordance with any SEC Restrictions applicable to such Cut Back Shares (such date, the “Restriction Termination Date”). From and after the Restriction Termination Date applicable to any Cut Back Shares, all of the provisions of this Section 2 (including the Company’s obligations with respect to the filing of a Registration Statement and its obligations to use reasonable efforts to have such Registration Statement declared effective within the time periods set forth herein and the liquidated damages provisions relating thereto) shall again be applicable to such Cut Back Shares; provided, however, that the date by which the Company is required to file the Registration Statement with respect to such Cut Back Shares shall be the tenth day following the Restriction Termination Date and the date by which the Company is required to have the Registration Statement effective with respect to such Cut Back Shares shall be the 55th day immediately after the Restriction Termination Date.
3. RELATED COMPANY OBLIGATIONS.
With respect to the Registration Statement and whenever any Registrable Securities are to be Registered pursuant to Section 2, including on the Initial Registration Statement or on any New Registration Statement, the Company shall use its reasonable best efforts to effect the registration of the Registrable Securities in accordance with the intended method of disposition thereof and, pursuant thereto, the Company shall have the following obligations:
(a) Notifications. The Company will promptly notify the Investors of the time when any subsequent amendment to the Initial Registration Statement or any New Registration Statement, other than documents incorporated by reference, has been filed with the SEC and/or has become effective or where a receipt has been issued therefor or any subsequent supplement to a prospectus has been filed and of any request by the SEC for any amendment or supplement to the Registration Statement, any New Registration Statement or any prospectus or for additional information.
(b) Amendments. The Company will prepare and file with the SEC any amendments, post-effective amendments or supplements to the Initial Registration Statement, any New Registration Statement or any related prospectus, as applicable, (a) as may be necessary to keep such Registration Statement effective for the Registration Period and to comply with the provisions of the Securities Act and the Securities Exchange Act of 1934, as amended (the “Exchange Act”) with respect to the distribution of all of the Registrable Securities covered thereby, or (b) in the reasonable opinion of the Investors and the Company, as may be necessary or advisable in connection with any acquisition or sale of Registrable Securities by the Investors.
(c) Investor Review. The Company will not file any amendment or supplement to the Registration Statement, any New Registration Statement or any prospectus, other than documents incorporated by reference, relating to the Investors, the Registrable Securities or the transactions contemplated hereby unless (A) the Investors and their counsel shall have been advised and afforded the opportunity to review and comment thereon at least three (3) Business Days prior to filing with the SEC
5
and (B) the Company shall have given reasonable due consideration to any comments thereon received from the Investors or their counsel.
(d) Copies Available. The Company will furnish to any Investor whose Registrable Securities are included in any Registration Statement and its counsel copies of the Initial Registration Statement, any prospectus thereunder (including all documents incorporated by reference therein), any prospectus supplement thereunder, any New Registration Statement and all amendments to the Initial Registration Statement or any New Registration Statement that are filed with the SEC during the Registration Period (including all documents filed with or furnished to the SEC during such period that are deemed to be incorporated by reference therein), each letter written by or on behalf of the Company to the SEC or the staff of the SEC, and each item of correspondence from the SEC or the staff of the SEC, in each case relating to such Registration Statement (other than any portion thereof which contains information for which the Company has sought confidential treatment) and such other documents as Investor may reasonably request in order to facilitate the disposition of the Registrable Securities owned by Investor that are covered by such Registration Statement, in each case as soon as reasonably practicable upon such Investor’s request and in such quantities as such Investor may from time to time reasonably request; provided, however, that the Company shall not be required to furnish any document to the Investor to the extent such document is available on EDGAR.
(e) Notification of Stop Orders; Material Changes. The Company shall use commercially reasonable efforts to (i) prevent the issuance of any stop order or other suspension of effectiveness and, (ii) if such order is issued, obtain the withdrawal of any such order as soon as practicable. The Company shall advise the Investors promptly (but in no event later than 24 hours) and shall confirm such advice in writing, in each case: (i) of the Company’s receipt of notice of any request by the SEC or any other federal or state governmental authority for amendment of or a supplement to the Registration Statement or any prospectus or for any additional information; (ii) of the Company’s receipt of notice of the issuance by the SEC or any other federal or state governmental authority of any stop order suspending the effectiveness of the Initial Registration Statement or prohibiting or suspending the use of any prospectus or prospectus supplement, or any New Registration Statement, or of the Company’s receipt of any notification of the suspension of qualification of the Registrable Securities for offering or sale in any jurisdiction or the initiation or contemplated initiation of any proceeding for such purpose; and (iii) of the Company becoming aware of the happening of any event, which makes any statement of a material fact made in any Registration Statement or any prospectus untrue or which requires the making of any additions to or changes to the statements then made in any Registration Statement or any prospectus in order to state a material fact required by the Securities Act to be stated therein or necessary in order to make the statements then made therein (in the case of any prospectus, in light of the circumstances under which they were made) not misleading, or of the necessity to amend any Registration Statement or any prospectus to comply with the Securities Act or any other law. The Company shall not be required to disclose to the Investors the substance of specific reasons of any of the events set forth in clause (i) to (iii) of the immediately preceding sentence (each, a “Suspension Event”), but rather, shall only be required to disclose that the event has occurred. If at any time the SEC, or any other federal or state governmental authority shall issue any stop order suspending the effectiveness of any Registration Statement or prohibiting or suspending the use of any prospectus or prospectus supplement, the Company shall use its reasonable best efforts to obtain the withdrawal of such order at the earliest practicable time. The Company shall furnish to the Investors, without charge, a copy of any correspondence from the SEC or the staff of the SEC, or any other federal or state governmental authority to the Company or its representatives relating to the Initial Registration Statement, any New Registration Statement or any prospectus, or prospectus supplement as the case may be. In the event of a Suspension Event set forth in
6
clause (iii) of the first sentence of this Section 3(e), the Company will use its commercially reasonable efforts to publicly disclose such event as soon as reasonably practicable, or otherwise resolve the matter such that sales under Registration Statements may resume; provided, however, that if the Company has a bona fide business purpose for not making such information public, the Company may suspend the use of all Registration Statements for up to 60 consecutive calendar days; provided, further, that the Company may not suspend the use of all Registration Statements more than twice, or for more than 90 total calendar days, in each case during any twelve-month period.
(f) Confirmation of Effectiveness. If reasonably requested by an Investor at any time in respect of any Registration Statement, the Company shall deliver to such Investor a written confirmation (email being sufficient) from Company’s counsel of whether or not the effectiveness of such Registration Statement has lapsed at any time for any reason (including, without limitation, the issuance of a stop order) and whether or not such Registration Statement is currently effective and available to the Company for sale of Registrable Securities.
(g) Listing. The Company shall use best efforts to cause all Registrable Securities covered by a Registration Statement to be listed on the Nasdaq Global Select Market.
(h) Compliance. The Company shall otherwise use best efforts to comply with all applicable rules and regulations of the SEC under the Securities Act and the Exchange Act, including, without limitation, Rule 172 under the Securities Act, file any final prospectus, including any supplement or amendment thereof, with the SEC pursuant to Rule 424 under the Securities Act, promptly inform the Investor in writing if, at any time during the Registration Period, the Company does not satisfy the conditions specified in Rule 172 and, as a result thereof, the Investor is required to deliver a prospectus in connection with any disposition of Registrable Securities and take such other actions as may be reasonably necessary to facilitate the registration of the Registrable Securities hereunder, and make available to its security holders, as soon as reasonably practicable, but not later than the Availability Date (as defined below), an earnings statement covering a period of at least 12 months, beginning after the effective date of each Registration Statement, which earnings statement shall satisfy the provisions of Section 11(a) of the Securities Act, including Rule 158 promulgated thereunder (for the purpose of this subsection 3(h), “Availability Date” means the 45th day following the end of the fourth fiscal quarter that includes the effective date of such Registration Statement, except that, if such fourth fiscal quarter is the last quarter of the Company’s fiscal year, “Availability Date” means the 90th day after the end of such fourth fiscal quarter).
(i) Blue-Sky. The Company shall register or qualify or cooperate with the Investor and their counsel in connection with the registration or qualification of such Registrable Securities for the offer and sale under the securities or blue sky laws of such jurisdictions reasonably requested by the Investor; provided, however, that the Company shall not be required in connection therewith or as a condition thereto to (i) qualify to do business in any jurisdiction where it would not otherwise be required to qualify but for this Section 3(i), (ii) subject itself to general taxation in any jurisdiction where it would not otherwise be so subject but for this Section 3(i), or (iii) file a general consent to service of process in any such jurisdiction.
(j) Rule 144. With a view to making available to the Investors the benefits of Rule 144 (or its successor rule) and any other rule or regulation of the SEC that may at any time permit the Investors to sell shares of Common Stock to the public without registration, the Company covenants and agrees to: (i) make and keep adequate current public information available, as those terms are understood and defined in Rule 144, until the earlier of (A) six months after such date as all of the
7
Registrable Securities may be sold without restriction by the holders thereof pursuant to Rule 144 or any other rule of similar effect or (B) such date as there are no longer Registrable Securities; (ii) file with the SEC in a timely manner all reports and other documents required of the Company under the Exchange Act; and (iii) furnish electronically to each Investor upon request, as long as such Investor owns any Registrable Securities, (A) a written statement by the Company that it has complied with the reporting requirements of the Exchange Act, (B) a copy of or electronic access to the Company’s most recent Annual Report on Form 10-K or Quarterly Report on Form 10-Q, and (C) such other information as may be reasonably requested in order to avail such Investor of any rule or regulation of the SEC that permits the selling of any such Registrable Securities without registration.
(k) Cooperation. The Company shall cooperate with the holders of the Registrable Securities to facilitate the timely preparation and delivery of certificates or uncertificated shares representing the Registrable Securities to be sold pursuant to such Registration Statement or Rule 144 free of any restrictive legends and representing such number of shares of Common Stock and registered in such names as the holders of the Registrable Securities may reasonably request to the extent permitted by such Registration Statement or Rule 144 to effect sales of Registrable Securities ; for the avoidance of doubt, the Company may satisfy its obligations hereunder without issuing physical stock certificates through the use of The Depository Trust Company’s Direct Registration System.
4. OBLIGATIONS OF THE INVESTORS.
(a) Investor Information. Each Investor shall provide a completed Investor Questionnaire in the form attached hereto as Exhibit B or such other form or information required by the Company in connection with the registration of the Registrable Securities. If the Company has not received such completed Questionnaire from an Investor within three business days of the Company’s request, the Company may file the Registration Statement without including such Investor’s Registrable Securities.
(b) Suspension of Sales. Each Investor, severally and not jointly with any other Investor, agrees that, upon receipt of any notice from the Company of the existence of Suspension Event as set forth in Section 3(e), the Investor will immediately discontinue disposition of Registrable Securities pursuant to any Registration Statement covering such Registrable Securities until the Investor's receipt of a notice from the Company confirming the resolution of such Suspension Event and that such dispositions may again be made.
(c) Investor Cooperation. Each Investor, severally and not jointly with any other Investor, agrees to cooperate with the Company as reasonably requested by the Company in connection with the preparation and filing of any amendments and supplements to any Registration Statement or New Registration Statement hereunder, unless such Investor has notified the Company in writing of its election to exclude all of its Registrable Securities from such Registration Statement.
5. EXPENSES OF REGISTRATION.
All Registration Expenses incurred in connection with registrations pursuant to this Agreement shall be borne by the Company. All Selling Expenses relating to securities registered on behalf of the Investors shall be borne by the Investors pro rata on the basis of the number of Registrable Securities so registered.
8
6. INDEMNIFICATION.
(a) To the fullest extent permitted by law, the Company will, and hereby does, indemnify, hold harmless and defend the Investors, each Person, if any, who controls the Investors, the members, the directors, officers, partners, employees, members, managers, agents, representatives and advisors of the Investors and each Person, if any, who controls the Investors within the meaning of the Securities Act or the Exchange Act (each, an “Indemnified Person”), against any losses, obligation, claims, damages, liabilities, contingencies, judgments, fines, penalties, charges, costs (including, without limitation, court costs and costs of preparation), reasonable and documented attorneys’ fees, amounts paid in settlement and reasonable and documented expenses (collectively, “Claims”)) reasonably incurred in investigating, preparing or defending any action, claim, suit, inquiry, proceeding, investigation or appeal taken from the foregoing by or before any court or governmental, administrative or other regulatory agency or body or the SEC, whether pending or threatened, whether or not an indemnified party is or may be a party thereto (“Indemnified Damages”), to which any of them may become subject insofar as such Claims (or actions or proceedings, whether commenced or threatened, in respect thereof) arise out of or are based upon: (i) any untrue statement or alleged untrue statement or omission or alleged omission of any material fact contained in any Registration Statement, any preliminary prospectus or final prospectus, or any amendment or supplement thereof, or (ii) any violation or alleged violation by the Company of the Securities Act, Exchange Act or any other state securities or other “blue sky” laws of any jurisdiction in which Registrable Securities are offered or any rule or regulation promulgated thereunder applicable to the Company or its agents and relating to action or inaction required of the Company in connection with such registration of the Registrable Securities (the matters in the foregoing clauses (i) and (ii) being, collectively, “Violations”). The Company shall reimburse each Indemnified Person promptly as such expenses are incurred and are due and payable, for any reasonable out of pocket legal fees or other reasonable and documented expenses incurred by them in connection with investigating or defending any such Claim. Notwithstanding anything to the contrary contained herein, the indemnification agreement contained in this Section 6(a): (A) shall not apply to a Claim by an Indemnified Person arising out of or based upon a Violation which occurs in reliance upon and in conformity with information furnished in writing to the Company by the relevant Investor or such relevant Indemnified Person specifically for use in such Registration Statement or prospectus and was reviewed and approved in writing by such Investor or such Indemnified Person expressly for use in connection with the preparation of any Registration Statement, any prospectus or any such amendment thereof or supplement thereto, if such in each case if the foregoing was timely made available by the Company; (B) with respect to any superseded prospectus, shall not inure to the benefit of any such Person from whom the Person asserting any such Claim purchased the Registrable Securities that are the subject thereof (or to the benefit of any other Indemnified Person) if the untrue statement or omission of material fact contained in the superseded prospectus was corrected in the revised prospectus, as then amended or supplemented, and the Indemnified Person was promptly advised in writing not to use the outdated, defective or incorrect prospectus prior to the use giving rise to a violation; (C) shall not apply to amounts paid in settlement of any Claim if such settlement is effected without the prior written consent of the Company, which consent shall not be unreasonably withheld, conditioned or delayed. Such indemnity shall remain in full force and effect regardless of any investigation made by or on behalf of the Indemnified Person and shall survive the transfer of the Registrable Securities by the Investor pursuant to Section 8.
(b) In connection with the Initial Registration Statement, any New Registration Statement or any prospectus, each Investor, severally and not jointly, agrees to indemnify, hold harmless and defend, the Company, each of its directors, each of its officers who signed the Initial Registration Statement or signs any New Registration Statement, and each Person, if any, who controls the Company within the meaning of the Securities Act or the Exchange Act (each, an “Indemnified Party”), against
9
any losses, claims, damages, liabilities and expense (including reasonable attorney fees) resulting from any Violation (as defined below), in each case to the extent, and only to the extent, that such Violation occurs in reliance upon and in conformity with information about the relevant Investor furnished in writing by such Investor to the Company for use in such Registration Statement and reviewed and approved in writing by such Investor or such Indemnified Person expressly for use in connection with the preparation of the Registration Statement, any New Registration Statement, any prospectus or any such amendment thereof or supplement thereto. In no event shall the liability of any Investor be greater in amount than the dollar amount of the proceeds (net of all expenses paid by such Investor in connection with any claim relating to this Section 6 and the amount of any damages such Investor has otherwise been required to pay by reason of such untrue statement or omission) received by such Investor upon the sale of the Registrable Securities included in such Registration Statement giving rise to such indemnification obligation. Such indemnity shall remain in full force and effect regardless of any investigation made by or on behalf of such Indemnified Party and shall survive the transfer of the Registrable Securities by any Investor pursuant to Section 8.
(c) Promptly after receipt by an Indemnified Person or Indemnified Party under this Section 6 of notice of the commencement of any action or proceeding (including any governmental action or proceeding) involving a Claim, such Indemnified Person or Indemnified Party shall, if a Claim in respect thereof is to be made against any indemnifying party under this Section 6, deliver to the indemnifying party a written notice of the commencement thereof, and the indemnifying party shall have the right to participate in, and, to the extent the indemnifying party so desires, jointly with any other indemnifying party similarly noticed, to assume control of the defense thereof with counsel mutually satisfactory to the indemnifying party and the Indemnified Person or the Indemnified Party, as the case may be, and upon such notice, the indemnifying party shall not be liable to the Indemnified Person or the Indemnified Party for any legal or other expenses subsequently incurred by the Indemnified Person or the Indemnified Party in connection with the defense thereof; provided, however, that an Indemnified Person or Indemnified Party (together with all other Indemnified Persons and Indemnified Parties that may be represented without conflict by one counsel) shall have the right to retain its own counsel with the reasonable fees and expenses to be paid by the indemnifying party, if, in the reasonable opinion of counsel retained by the indemnifying party, the representation by such counsel of the Indemnified Person or Indemnified Party and the indemnifying party would be inappropriate due to actual or potential differing interests between such Indemnified Person or Indemnified Party and any other party represented by such counsel in such proceeding. The Indemnified Party or Indemnified Person shall cooperate with the indemnifying party in connection with any negotiation or defense of any such action or claim by the indemnifying party and shall furnish to the indemnifying party all information reasonably available to the Indemnified Party or Indemnified Person which relates to such action or claim. The indemnifying party shall keep the Indemnified Party or Indemnified Person fully apprised as to the status of the defense or any settlement negotiations with respect thereto. No indemnifying party shall be liable for any settlement of any action, claim or proceeding effected without its written consent, provided, however, that the indemnifying party shall not unreasonably withhold, delay or condition its consent. No indemnifying party shall, without the consent of the Indemnified Party or Indemnified Person, consent to entry of any judgment or enter into any settlement or other compromise unless such judgment or settlement (i) imposes no liability or obligation on, (ii) includes as an unconditional term thereof the giving of a complete, explicit and unconditional release from the party bringing such indemnified claims of all liability of the Indemnified Party or Indemnified Person in respect to or arising out of such claim or litigation in favor of, and (iii) does not include any admission of fault, culpability, wrongdoing, or wrongdoing or malfeasance by or on behalf of, the Indemnified Party or Indemnified Person. Following indemnification as provided for hereunder, the indemnifying party shall be subrogated to all rights of the Indemnified Party or Indemnified Person with respect to all third parties, firms or corporations relating to
10
the matter for which indemnification has been made. The failure to deliver written notice to the indemnifying party within a reasonable time of the commencement of any such action shall not relieve such indemnifying party of any liability to the Indemnified Person or Indemnified Party under this Section 6, except to the extent that the indemnifying party is prejudiced in its ability to defend such action.
(d) The indemnification required by this Section 6 shall be made by periodic payments of the amount thereof during the course of the investigation or defense, as and when bills are received or Indemnified Damages are incurred. Any Person receiving a payment pursuant to this Section 6 which person is later determined to not be entitled to such payment shall return such payment (including reimbursement of expenses) to the person making it.
(e) The indemnity agreements contained herein shall be in addition to (i) any cause of action or similar right of the Indemnified Party or Indemnified Person against the indemnifying party or others, and (ii) any liabilities the indemnifying party may be subject to pursuant to the law.
7. CONTRIBUTION.
To the extent any indemnification by an indemnifying party is prohibited or limited by law, the indemnifying party agrees to make the maximum contribution with respect to any amounts for which it would otherwise be liable under Section 6 to the fullest extent permitted by law; provided, however, that: (i) no seller of Registrable Securities guilty of fraudulent misrepresentation (within the meaning of Section 11(f) of the Securities Act) shall be entitled to contribution from any seller of Registrable Securities who was not guilty of fraudulent misrepresentation; and (ii) contribution by any seller of Registrable Securities shall be limited in amount to the net amount of proceeds (net of all expenses paid by such holder in connection with any claim relating to this Section 7 and the amount of any damages such holder has otherwise been required to pay by reason of such untrue or alleged untrue statement or omission or alleged omission) received by such seller from the sale of such Registrable Securities giving rise to such contribution obligation.
8. ASSIGNMENT OF REGISTRATION RIGHTS.
The Company shall not assign this Agreement or any rights or obligations hereunder (whether by operation of law or otherwise) without the prior written consent of the Investors holding a majority of the Registrable Securities then outstanding; provided, however, that in any transaction, whether by merger, reorganization, restructuring, consolidation, financing or otherwise, whereby the Company is a party and in which the Registrable Securities are converted into the equity securities of another Person, from and after the effective time of such transaction, such Person shall, by virtue of such transaction, be deemed to have assumed the obligations of the Company hereunder, the term “Company” shall be deemed to refer to such Person and the term “Registrable Securities” shall be deemed to include the securities received by the Investor in connection with such transaction unless such securities are otherwise freely tradable by the Investor after giving effect to such transaction, and the prior written consent of the Investors holding a majority of the Registrable Securities then outstanding shall not be required for such transaction.
An Investor may transfer or assign its rights hereunder, in whole or from time to time in part, to one or more Persons in connection with the transfer of Registrable Securities by such Investor to such Person, provided that such Investor complies with all laws applicable thereto, and the provisions of the Purchase Agreement, and provides written notice of assignment to the Company promptly after such
11
assignment is effected, and such Person agrees in writing to be bound by all of the provisions contained herein.
The provisions of this Agreement shall be binding upon and inure to the benefit of the Investor and its successors and permitted assigns.
9. AMENDMENTS AND WAIVERS.
The provisions of this Agreement, including the provisions of this sentence, may be amended, modified or supplemented, or waived only by a written instrument executed by (i) the Company and (ii) the holders of a majority of the then outstanding Registrable Securities, provided that (A) any party may give a waiver as to itself, and (B) any amendment, modification, supplement or waiver that disproportionately and adversely affects the rights and obligations of any Investor relative to the comparable rights and obligations of the other Investors shall require the prior written consent of such adversely affected Investor or each Investor, as applicable. Notwithstanding the foregoing, a waiver or consent to depart from the provisions hereof with respect to a matter that relates exclusively to the rights of one or more Investors and that does not adversely directly or indirectly affect the rights of other Investors may be given by Investors holding all of the Registrable Securities to which such waiver or consent relates.
10. MISCELLANEOUS.
(a) Notices. Any notices or other communications required or permitted to be given hereunder shall be in writing and shall be deemed to be given (a) when delivered if personally delivered to the party for whom it is intended, (b) when delivered, if sent by electronic mail during normal business hours of the recipient, and if not sent during normal business hours, then on the recipient’s next business day, (c) three days after having been sent by certified or registered mail, return-receipt requested and postage prepaid, or (d) one business day after deposit with a nationally recognized overnight courier, freight prepaid, specifying next business day delivery, with written verification of receipt:
i. If to the Company, addressed as follows:
Acumen Pharmaceuticals, Inc.
1210-1220 Washington St., Suite 210
Newton, MA 02465
Attention: Derek Meisner
Email:
with a copy (which shall not constitute notice):
Ropes & Gray LLP
Prudential Tower, 800 Boylston Street
Boston, MA 02199-3600
Attention: Thomas Danielski
Email:
ii. If to any Investor, at its e-mail address or address set forth on its signature page to the Purchase Agreement or to such e-mail address, or address as subsequently modified by written notice given in accordance with this Section 10.
12
Any Person may change the address to which notices and communications to it are to be addressed by notification as provided for herein.
(b) Consent to Electronic Notice. Each Investor consents to the delivery of any stockholder notice pursuant to the Delaware General Corporation Law (the “DGCL”), as amended or superseded from time to time, by electronic mail pursuant to Section 232 of the DGCL (or any successor thereto) at the e-mail address set forth below the Investor’s name on the signature page or Exhibit A, as updated from time to time by notice to the Company. To the extent that any notice given by means of electronic mail is returned or undeliverable for any reason, the foregoing consent shall be deemed to have been revoked until a new or corrected e-mail address has been provided, and such attempted electronic notice shall be ineffective and deemed to not have been given. Each party agrees to promptly notify the other parties of any change in its e-mail address, and that failure to do so shall not affect the foregoing.
(c) Waiver. No waiver of any term, provision or condition of this Agreement, whether by conduct or otherwise, in any one or more instances, shall be deemed to be, or be construed as, a further or continuing waiver of any such term, provision or condition or as a waiver of any other term, provision or condition of this Agreement.
(d) Governing Law. The provisions of Section 8.5 of the Purchase Agreement are incorporated by reference herein mutatis mutandis.
(e) Headings. The titles, subtitles and headings in this Agreement are for convenience of reference and shall not form part of, or affect the interpretation of, this Agreement.
(f) Counterparts. This Agreement may be executed in two or more identical counterparts, all of which shall be considered one and the same agreement and shall become effective when counterparts have been signed by each party and delivered to the other party; provided that a facsimile or pdf signature including any electronic signatures complying with the U.S. federal ESIGN Act of 2000, e.g., www.docusign.com shall be considered due execution and shall be binding upon the signatory thereto with the same force and effect as if the signature were an original, not a facsimile or pdf (or other electronic reproduction) of a signature.
(g) Further Assurances. Each party shall do and perform, or cause to be done and performed, all such further acts and things, and shall execute and deliver all such other agreements, certificates, instruments and documents as the other party may reasonably request in order to carry out the intent and accomplish the purposes of this Agreement and the consummation of the transactions contemplated hereby.
(h) Contract Interpretation. This Agreement is the joint product of each Investor and the Company and each provision hereof has been subject to the mutual consultation, negotiation and agreement of such parties and shall not be construed for or against any party hereto.
(i) No Third Party Beneficiaries. Nothing in this Agreement, express or implied, is intended to confer on any Person other than the parties to this Agreement any rights, remedies, claims, benefits, obligations or liabilities under or by reason of this Agreement, and no Person that is not a party to this Agreement (including, without limitation, any partner, member, shareholder, director, officer, employee or other beneficial owner of any party to this Agreement, in its own capacity as such or in
13
bringing a derivative action on behalf of a party to this Agreement) shall have any standing as a third party beneficiary with respect to this Agreement or the transactions contemplated hereby.
(j) Severability. If any part or provision of this Agreement is held unenforceable or in conflict with the applicable laws or regulations of any jurisdiction, the invalid or unenforceable part or provisions shall be replaced with a provision which accomplishes, to the extent possible, the original business purpose of such part or provision in a valid and enforceable manner, and the remainder of this Agreement shall remain binding upon the parties hereto.
(k) Non-Recourse. Notwithstanding anything that may be expressed or implied in this Agreement, the Company covenants, agrees and acknowledges that no recourse under this Agreement or any documents or instruments delivered in connection with this Agreement shall be had against any current or future director, officer, employee, stockholder, general or limited partner or member of the Investors or of any affiliates or assignees thereof, whether by the enforcement of any assessment or by any legal or equitable proceeding, or by virtue of any statute, regulation or other applicable law, it being expressly agreed and acknowledged that no personal liability whatsoever shall attach to, be imposed on or otherwise be incurred by any current or future director, officer, employee, stockholder, general or limited partner or member of the Investors or of any affiliates or assignees thereof, as such for any obligation of the Investors under this Agreement or any documents or instruments delivered in connection with this Agreement for any claim based on, in respect of or by reason of such obligations or their creation.
(l) Specific Performance. In addition to any and all other remedies that may be available at law in the event of any breach of this Agreement, each Investor shall be entitled to specific performance of the agreements and obligations of the Company hereunder and to such other injunction or other equitable relief as may be granted by a court of competent jurisdiction.
(m) Cumulative Remedies. The remedies provided herein are cumulative and not exclusive of any remedies provided by law.
[Signature Page Follows]
14
IN WITNESS WHEREOF, the parties have caused this Registration Rights Agreement to be duly executed as of date first written above.
COMPANY:
ACUMEN PHARMACEUTICALS, INC.
ACUMEN PHARMACEUTICALS, INC.
By: __________________________
Name: Daniel O’Connell
Title: Chief Executive Officer
Name: Daniel O’Connell
Title: Chief Executive Officer
[Signature Page to Registration Rights Agreement]
IN WITNESS WHEREOF, the parties have caused this Registration Rights Agreement to be duly executed as of date first written above.
INVESTOR:
By: ___________________
Name: _________________
Title: __________________
[Signature Page to Registration Rights Agreement]
Exhibit A
PLAN OF DISTRIBUTION
The selling stockholders, which as used herein includes donees, pledgees, transferees or other successors-in-interest selling shares of common stock or interests in shares of common stock received after the date of this prospectus from a selling stockholder as a gift, pledge, partnership distribution or other transfer, may, from time to time, sell, transfer or otherwise dispose of any or all of their shares of common stock or interests in shares of common stock on any stock exchange, market or trading facility on which the shares are traded or in private transactions. These dispositions may be at fixed prices, at prevailing market prices at the time of sale, at prices related to the prevailing market price, at varying prices determined at the time of sale, or at negotiated prices.
The selling stockholders may use any one or more of the following methods when disposing of shares or interests therein:
• distributions to members, partners, stockholders or other equityholders of the selling stockholders;
• ordinary brokerage transactions and transactions in which the broker-dealer solicits purchasers;
• block trades in which the broker-dealer will attempt to sell the shares as agent, but may position and resell a portion of the block as principal to facilitate the transaction;
• purchases by a broker-dealer as principal and resale by the broker-dealer for its account;
• an exchange distribution in accordance with the rules of the applicable exchange;
• privately negotiated transactions;
• short sales and settlement of short sales entered into after the effective date of the registration statement of which this prospectus is a part;
• through the writing or settlement of options or other hedging transactions, whether through an options exchange or otherwise;
• broker-dealers may agree with the selling stockholders to sell a specified number of such shares at a stipulated price per share;
• a combination of any such methods of sale; and
• any other method permitted pursuant to applicable law.
The selling stockholders may, from time to time, pledge or grant a security interest in some or all of the shares of common stock owned by them and, if they default in the performance of their secured obligations, the pledgees or secured parties may offer and sell the shares of common stock, from time to time, under this prospectus, or under an amendment to this prospectus under Rule 424(b)(3) or other applicable provision of the Securities Act, amending the list of selling stockholders to include the
1
pledgee, transferee or other successors in interest as selling stockholders under this prospectus. The selling stockholders also may transfer the shares of common stock in other circumstances, in which case the transferees, pledgees or other successors in interest will be the selling stockholders for purposes of this prospectus.
In connection with the sale of our common stock or interests therein, the selling stockholders may enter into hedging transactions with broker-dealers or other financial institutions, which may in turn engage in short sales of the common stock in the course of hedging the positions they assume. The selling stockholders may also sell shares of our common stock short and deliver these securities to close out their short positions, or loan or pledge the common stock to broker-dealers that in turn may sell these securities. The selling stockholders may also enter into option or other transactions with broker-dealers or other financial institutions or the creation of one or more derivative securities which require the delivery to such broker-dealer or other financial institution of shares offered by this prospectus, which shares such broker-dealer or other financial institution may resell pursuant to this prospectus (as supplemented or amended to reflect such transaction).
The aggregate proceeds to the selling stockholders from the sale of the common stock offered by them will be the purchase price of the common stock less discounts or commissions, if any. Each of the selling stockholders reserves the right to accept and, together with their agents from time to time, to reject, in whole or in part, any proposed purchase of common stock to be made directly or through agents. We will not receive any of the proceeds from this offering.
The selling stockholders also may resell all or a portion of the shares in open market transactions in reliance upon Rule 144 under the Securities Act, provided that they meet the criteria and conform to the requirements of that rule, or another available exemption from the registration requirements under the Securities Act.
The selling stockholders and any underwriters, broker-dealers or agents that participate in the sale of the common stock or interests therein may be “underwriters” within the meaning of Section 2(a)(11) of the Securities Act (it being understood that the selling stockholders shall not be deemed to be underwriters solely as a result of their participation in this offering). Any discounts, commissions, concessions or profit they earn on any resale of the shares may be underwriting discounts and commissions under the Securities Act. Selling stockholders who are “underwriters” within the meaning of Section 2(a)(11) of the Securities Act will be subject to the prospectus delivery requirements of the Securities Act.
To the extent required, the shares of our common stock to be sold, the names of the selling stockholders, the respective purchase prices and public offering prices, the names of any agent, dealer or underwriter, and any applicable commissions or discounts with respect to a particular offer will be set forth in an accompanying prospectus supplement or, if appropriate, a post-effective amendment to the registration statement that includes this prospectus.
In order to comply with the securities laws of some states, if applicable, the common stock may be sold in these jurisdictions only through registered or licensed brokers or dealers. In addition, in some states the common stock may not be sold unless it has been registered or qualified for sale or an exemption from registration or qualification requirements is available and is complied with.
2
We have advised the selling stockholders that the anti-manipulation rules of Regulation M under the Exchange Act may apply to sales of shares in the market and to the activities of the selling stockholders and their affiliates. In addition, to the extent applicable, we will make copies of this prospectus (as it may be supplemented or amended from time to time) available to the selling stockholders for the purpose of satisfying the prospectus delivery requirements of the Securities Act. The selling stockholders may indemnify any broker-dealer that participates in transactions involving the sale of the shares against certain liabilities, including liabilities arising under the Securities Act.
We have agreed to indemnify the selling stockholders against liabilities, including liabilities under the Securities Act and state securities laws, relating to the registration of the shares offered by this prospectus.
We have agreed with the selling stockholders to use commercially reasonable efforts to cause the registration statement of which this prospectus constitutes a part to become effective and to remain continuously effective until the earlier of: (i) the date on which the selling stockholders shall have resold or otherwise disposed of all the shares covered by this prospectus and (ii) the date on which the shares covered by this prospectus no longer constitute “Registrable Securities” as such term is defined in the Registration Rights Agreement, such that they may be resold by the selling stockholders without registration and without regard to any volume or manner-of-sale limitations and without current public information pursuant to Rule 144 under the Securities Act or any other rule of similar effect.
3
Exhibit B
Investor Questionnaire
The undersigned hereby provides the following information to the Company and represents and warrants that such information is accurate:
QUESTIONNAIRE
1. Name.
(a) Full Legal Name of Investor
(b) Full Legal Name of Registered Holder (if not the same as (a) above) through which Registrable Securities are held:
(c) Full Legal Name of Natural Control Person (which means a natural person who directly or indirectly alone or with others has power to vote or dispose of the securities covered by this Questionnaire):
2. Address for Notices to Investor:
Telephone: | ||
E-Mail: _____________________________________________________________________ | ||
Contact Person: | ||
4
3. Broker-Dealer Status:
(a) Are you a broker-dealer?
Yes No
(b) If “yes” to Section 3(a), did you receive your Registrable Securities as compensation for investment banking services to the Company?
Yes No
Note: If “no” to Section 3(b), the Commission’s staff has indicated that you should be identified as an underwriter in the Registration Statement.
(c) Are you an affiliate of a broker-dealer?
Yes No
(d) If you are an affiliate of a broker-dealer, do you certify that you purchased the Registrable Securities in the ordinary course of business, and at the time of the purchase of the Registrable Securities to be resold, you had no agreements or understandings, directly or indirectly, with any person to distribute the Registrable Securities?
Yes No
Note: If “no” to Section 3(d), the Commission’s staff has indicated that you should be identified as an underwriter in the Registration Statement.
4. Beneficial Ownership of Securities of the Company Owned by the Investor.
Except as set forth below in this Item 4, the undersigned is not the beneficial or registered owner of any securities of the Company other than the securities issuable pursuant to the Purchase Agreement.
(a) Type and Amount of other securities beneficially owned by the Investor:
5. Relationships with the Company:
Except as set forth below, neither the undersigned nor any of its affiliates, officers, directors or principal equity holders (owners of 5% of more of the equity securities of the undersigned) has held any position or office or has had any other material relationship with the Company (or its predecessors or affiliates) during the past three years.
5
State any exceptions here:
The undersigned agrees to promptly notify the Company of any material inaccuracies or changes in the information provided herein that may occur subsequent to the date hereof at any time while the Registration Statement remains effective; provided, that the undersigned shall not be required to notify the Company of any changes to the number of securities held or owned by the undersigned or its affiliates.
By signing below, the undersigned consents to the disclosure of the information contained herein in its answers to Items 1 through 5 and the inclusion of such information in the Registration Statement and the related prospectus and any amendments or supplements thereto. The undersigned understands that such information will be relied upon by the Company in connection with the preparation or amendment of the Registration Statement and the related prospectus and any amendments or supplements thereto.
IN WITNESS WHEREOF the undersigned, by authority duly given, has caused this Notice and Questionnaire to be executed and delivered either in person or by its duly authorized agent.
Date: Beneficial Owner:
By:
Name:
Title:
PLEASE EMAIL A .PDF COPY OF THE COMPLETED AND EXECUTED QUESTIONNAIRE TO:
6
Exhibit 99.1

Acumen Pharmaceuticals Announces $35.75 Million Private Placement to Advance Potential Best-in-Class Molecules from Amyloid Beta Oligomer-Selective Enhanced Brain Delivery Portfolio
•Financing demonstrates conviction in oligomer-selective approach and follows strong preclinical data, including in vitro, in vivo and non-human primate study results, that support development candidates in Acumen’s transferrin-receptor targeting Enhanced Brain Delivery (EBDTM) program
•Candidates exceeded key preclinical criteria, demonstrating elevated brain exposure in non-human primates up to 40-fold over native antibodies, low risk of anemia, and robust stability profiles to enable subcutaneous administration
•Lead clinical candidate IND targeted for mid-2027
NEWTON, Mass., Mar. 16, 2026 – Acumen Pharmaceuticals, Inc. (NASDAQ: ABOS) (“Acumen” or the “Company”), a clinical-stage biopharmaceutical company developing novel therapeutics that target toxic soluble amyloid b oligomers (AβOs) for the treatment of Alzheimer’s disease (AD), today announced that it has entered into a securities purchase agreement with certain institutional and accredited investors for a private placement of approximately $35.75 million of shares of its common stock at a price of $3.30 per share. Acumen expects to receive gross proceeds from the offering of approximately $35.75 million, before deducting offering expenses.
“This financing from committed institutional investors strongly validates our portfolio and our Enhanced Brain Delivery strategy,” said Daniel O’Connell, Chief Executive Officer of Acumen. “The transaction also underscores confidence in the potential value we are building with amyloid b oligomer-targeted antibodies for Alzheimer’s patients, caregivers and stakeholders in alignment with Acumen’s vision.”
The private placement was led by existing investor RA Capital Management, with participation from other investors, including ADAR1 Capital Management, Sands Capital, and a large investment management firm.
The private placement is expected to close on or about March 16, 2026, subject to the satisfaction of customary closing conditions.

Proceeds from the financing are expected to primarily support Acumen’s Enhanced Brain Delivery (EBD) program, including ongoing preclinical development work to support the nomination of a lead clinical candidate molecule, and for working capital and other general corporate purposes. Submission of an IND with respect to a lead clinical candidate is targeted for mid-2027.
The securities to be sold in the private placement have not been registered under the Securities Act of 1933, as amended (the “Securities Act”), or any state or other applicable jurisdictions’ securities laws, and may not be offered or sold in the United States absent registration or an applicable exemption from the registration requirements of the Securities Act and applicable state or other jurisdictions’ securities laws. Acumen has agreed to file a registration statement with the United States Securities and Exchange Commission (the “SEC”) registering the resale of the shares of common stock issued in the private placement, no later than two business days after the filing of its annual report on Form 10-K for the fiscal year ended December 31, 2025.
This press release shall not constitute an offer to sell or the solicitation of an offer to buy these securities, nor shall there be any offer, solicitation or sale of these securities in any jurisdiction in which such offer, solicitation or sale would be unlawful. Any offering of the securities under the resale registration statement will only be made by means of a prospectus.
EBD Non-Human Primate (NHP) Study Results
On March 16, 2026, Acumen also announced certain preclinical data, including in vitro, in vivo and NHP study results, that support development candidates in Acumen’s EBD program, conducted utilizing JCR Pharmaceuticals’ (“JCR”) blood-brain barrier penetrating technology, J-Brain Cargo®. The development candidate results are consistent and predictive across in vitro and in vivo studies, including murine and NHP data. Findings include:
Enhanced Brain Penetration
o Development candidates achieved 14-40x higher brain levels in NHPs compared to native antibodies at 24 hours
Low Anemia Risk
o Hematology data in NHPs indicate low potential for anemia with no adverse effects observed
Subcutaneous Dosing Capability
o Favorable stability profile and enhanced brain delivery support subcutaneous administration with low-volume devices

Acumen’s EBDTM program is part of an ongoing collaboration between Acumen and JCR announced in July 2025. J-Brain Cargo® technology is JCR’s proprietary drug delivery system that efficiently delivers drugs to target tissues, including the central nervous system, through receptor-mediated transcytosis. It is applicable to various modalities including antibodies, enzymes, oligonucleotides, lipid nanoparticles, gene and cell therapy, peptides and decoy receptors.
“We are very pleased with the preclinical EBD candidate profiles generated to date and impressed with the level of expertise contributed by our partner to ensure we exceeded our target profile for the program,” said Jim Doherty, President and Chief Development Officer of Acumen. “Candidates have shown robust in vivo brain penetration in multiple species, hematology endpoints in non-human primates suggestive of a low potential for anemia risk, and stability results supportive of subcutaneous dosing. Ours is the only EBD program in the anti-amyloid space that pairs an Aβ oligomer-targeting antibody with a validated TfR-targeting antibody technology, underscoring an exciting opportunity to develop a potential best-in-class treatment for people living with Alzheimer’s Disease.”
About Acumen Pharmaceuticals, Inc.
Acumen Pharmaceuticals is a clinical-stage biopharmaceutical company developing a novel therapeutic that targets toxic soluble amyloid beta oligomers (AβOs) for the treatment of Alzheimer’s disease (AD). Acumen’s scientific founders pioneered research on AβOs, which a growing body of evidence indicates are early and persistent triggers of Alzheimer’s disease pathology. Acumen is currently focused on advancing its lead investigational product candidate, sabirnetug (ACU193), a humanized monoclonal antibody that selectively targets toxic soluble AβOs, in its ongoing Phase 2 clinical trial ALTITUDE-AD (NCT06335173) in early symptomatic Alzheimer’s disease patients, following positive results in its Phase 1 trial INTERCEPT-AD. Acumen is also investigating a subcutaneous formulation of sabirnetug using Halozyme’s proprietary ENHANZE® drug delivery technology. Acumen is also collaborating with JCR Pharmaceuticals to develop an Enhanced Brain Delivery (EBD™) therapy for Alzheimer’s disease utilizing a transferrin-receptor-targeting blood-brain barrier-penetrating technology. The company is headquartered in Newton, Mass.
About the J-Brain Cargo® Platform Technology
JCR Pharmaceuticals has developed a proprietary blood-brain barrier (BBB)-penetrating technology, J-Brain Cargo®, to bring biotherapeutics into the central nervous system (CNS). The first drug developed based on this technology is IZCARGOTM (INN: pabinafusp alfa) and is approved in Japan for the treatment of a lysosomal storage disorder.

About JCR Pharmaceuticals Co., Ltd.
JCR Pharmaceuticals Co., Ltd. is a global specialty pharmaceutical company that develops treatments that go beyond rare diseases to solve the world’s most complex healthcare challenges. JCR continues to build upon our 50-year legacy in Japan while expanding our global footprint into the US, Europe, and Latin America. JCR’s innovative therapies address conditions like growth disorder, MPS II, Fabry disease, acute graft-versus-host disease, and renal anemia. JCR is also developing treatments for rare diseases like MPS I, MPS II, MPS IIIA and B, and more.
Forward-Looking Statements
Forward-Looking Statements
This press release contains forward-looking statements within the meaning of The Private Securities Litigation Reform Act of 1995. Any statement describing Acumen’s goals, expectations, financial or other projections, intentions or beliefs is a forward-looking statement and should be considered an at-risk statement. Words such as “believes,” “expects,” “anticipates,” “could,” “should,” “would,” “seeks,” “aims,” “plans,” “potential,” “will,” “milestone” and similar expressions are intended to identify forward-looking statements, although not all forward-looking statements contain these identifying words. Forward-looking statements include statements concerning the expected gross proceeds, use of proceeds and anticipated timing for closing of Acumen’s private placement, as well as Acumen’s business, the results and significance of Acumen's preclinical data that support development candidates in the Company’s EBD program, and Acumen’s plans to develop a candidate to treat Alzheimer's Disease utilizing EBD technology, including its expectations with respect to timing for the submission of an IND, as well as its potential for developing a best-in-class therapeutic candidate for people living with Alzheimer’s Disease. These statements are based upon the current beliefs and expectations of Acumen management, and are subject to certain factors, risks and uncertainties, particularly those inherent in the process of discovering, developing and commercializing safe and effective human therapeutics. Such risks may be amplified by the impacts of geopolitical events and macroeconomic conditions, such as rising inflation and interest rates, supply disruptions and uncertainty of credit and financial markets. These and other risks concerning Acumen’s programs are described in additional detail in Acumen’s filings with the Securities and Exchange Commission (“SEC”), including in Acumen’s most recent Annual Report on Form 10-K, and in subsequent filings with the SEC. Copies of these and other documents are available from Acumen. Additional information will be made available in other filings that Acumen makes from time to time with the SEC. These forward-looking statements speak only as of the date hereof, and Acumen expressly disclaims any obligation to update or revise any forward-looking statement, except as otherwise required by law, whether, as a result of new information, future events or otherwise.

CONTACTS:
Investors:
Alex Braun
abraun@acumenpharm.com
Media:
ICR Healthcare
AcumenPR@icrhealthcare.com
ICR Healthcare
AcumenPR@icrhealthcare.com

Corporate Presentation March 2026 1

Forward-Looking Statements 2 This presentation contains forward-looking statements within the meaning of The Private Securities Litigation Reform Act of 1995. Any statement describing Acumen’s goals, expectations, financial or other projections, intentions or beliefs is a forward-looking statement and should be considered an at-risk statement. Words such as “believes,” “expects,” “anticipates,” “could,” “would,” “seeks,” “aims,” “plans,” “potential,” “will” and similar expressions are intended to identify forward-looking statements, although not all forward-looking statements contain these identifying words. Forward-looking statements include statements concerning Acumen’s business, and Acumen’s ability to achieve its strategic and financial goals, including its projected use of cash, cash equivalents and marketable securities and the expected sufficiency of its cash resources into early 2027, the therapeutic potential of Acumen’s product candidate, sabirnetug (ACU193), including against other antibodies, the timing of anticipated topline results of ALTITUDE-AD, the potential for additional development to support a subcutaneous dosing option of sabirnetug, and the potential to develop a candidate to treat Alzheimer's Disease utilizing EBD technology. These statements are based upon the current beliefs and expectations of Acumen management, and are subject to certain factors, risks and uncertainties, particularly those inherent in the process of discovering, developing and commercializing safe and effective human therapeutics. Such risks may be amplified by the impacts of the COVID-19 pandemic. These and other risks concerning Acumen’s programs are described in additional detail in Acumen’s filings with the Securities and Exchange Commission (“SEC”), including in Acumen’s most recent Annual Report Form 10-K and future filings and reports by Acumen. Copies of these and other documents are available from Acumen. Additional information will be made available in other filings that Acumen makes from time to time with the SEC. These forward- looking statements speak only as of the date hereof, and Acumen expressly disclaims any obligation to update or revise any forward- looking statement, except as otherwise required by law, whether, as a result of new information, future events or otherwise. In this presentation, references to cash also include cash equivalents.
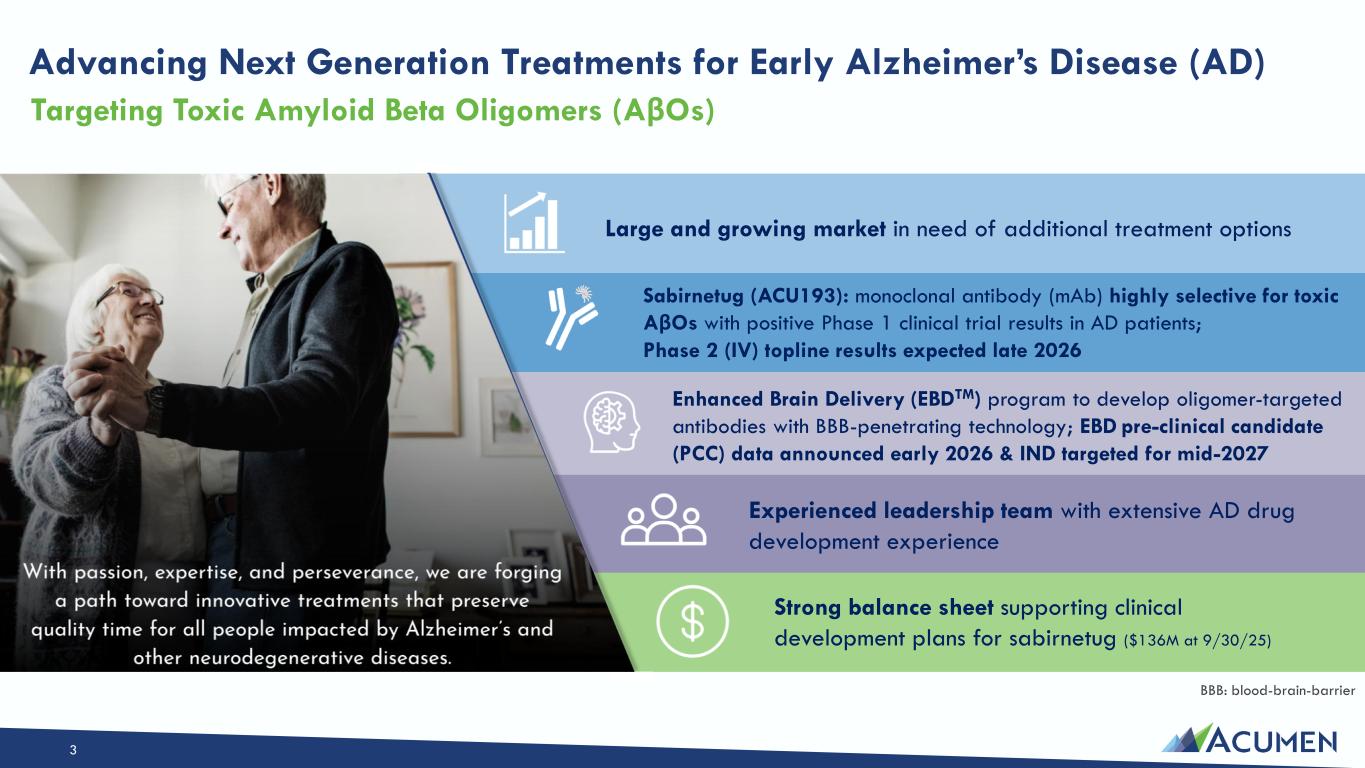
3 Large and growing market in need of additional treatment options Sabirnetug (ACU193): monoclonal antibody (mAb) highly selective for toxic AβOs with positive Phase 1 clinical trial results in AD patients; Phase 2 (IV) topline results expected late 2026 Enhanced Brain Delivery (EBDTM) program to develop oligomer-targeted antibodies with BBB-penetrating technology; EBD pre-clinical candidate (PCC) data announced early 2026 & IND targeted for mid-2027 Experienced leadership team with extensive AD drug development experience Strong balance sheet supporting clinical development plans for sabirnetug ($136M at 9/30/25) BBB: blood-brain-barrier Advancing Next Generation Treatments for Early Alzheimer’s Disease (AD) Targeting Toxic Amyloid Beta Oligomers (AβOs)
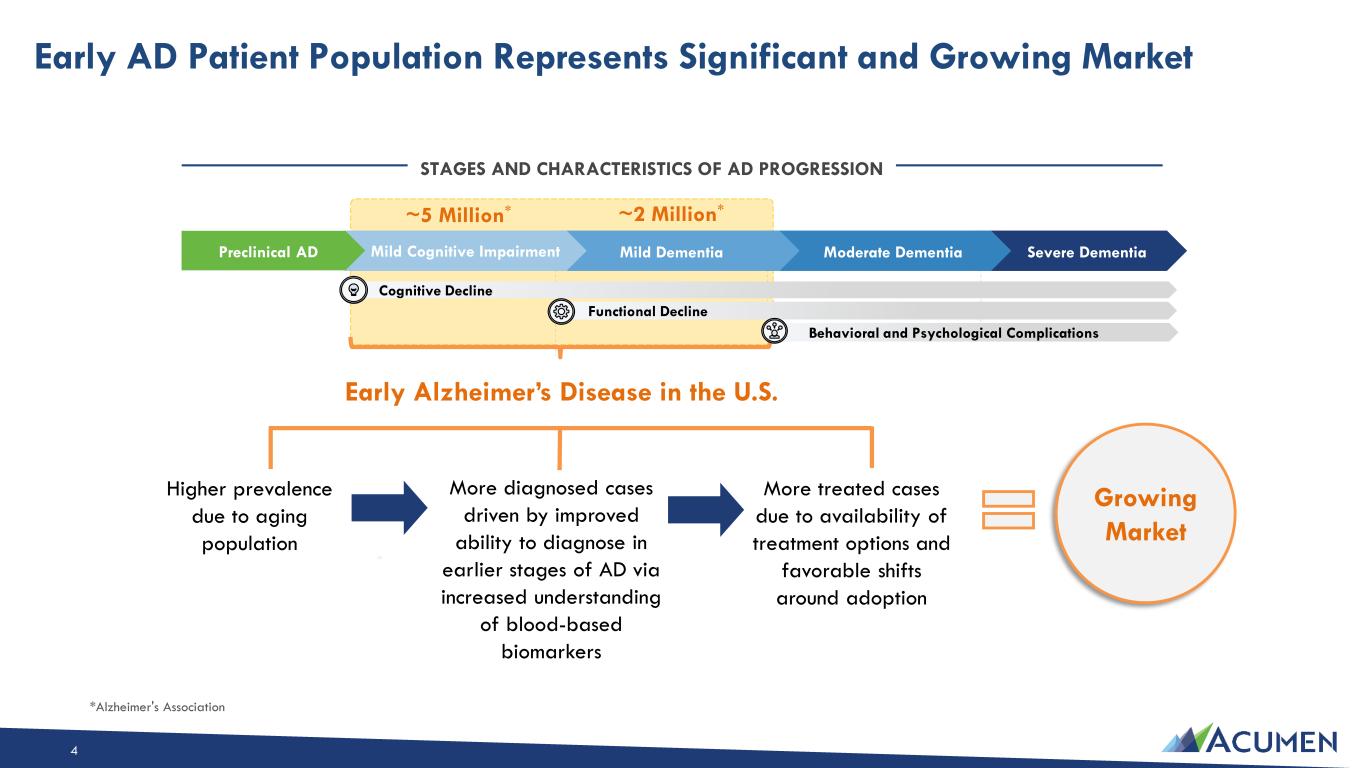
Early AD Patient Population Represents Significant and Growing Market STAGES AND CHARACTERISTICS OF AD PROGRESSION Severe DementiaModerate DementiaMild DementiaMild Cognitive ImpairmentPreclinical AD ~5 Million* ~2 Million* Early Alzheimer’s Disease in the U.S. Cognitive Decline Behavioral and Psychological Complications Functional Decline Growing Market Higher prevalence due to aging population More diagnosed cases driven by improved ability to diagnose in earlier stages of AD via increased understanding of blood-based biomarkers More treated cases due to availability of treatment options and favorable shifts around adoption *Alzheimer's Association 4

5 *ARIA-E: Amyloid-related imaging abnormalities - edema AD Treatment Landscape Continues to Evolve Approved Therapies Establish Momentum and Next-generation Approaches Expand Impact Future of AD Landscape Broadens Growth Prospects • New blood-based biomarkers and imaging tools will improve diagnostic and monitoring capabilities • Expansion into preclinical AD will significantly increase the addressable population • Combinations with other targets, such as tau, hold promise Current AD Treatments Offer Hope for People Living with AD • First approved anti-amyloid therapies steadily growing • Education and infrastructure buildout allowing for greater access to treatments by the addressable patient population • Increase efficacy • Reduce ARIA-E* risk • Provide more convenient delivery options • Target amyloid that occurs earlier in disease course Opportunities for New Anti-amyloid Therapies to Strengthen Risk-Benefit Tradeoff
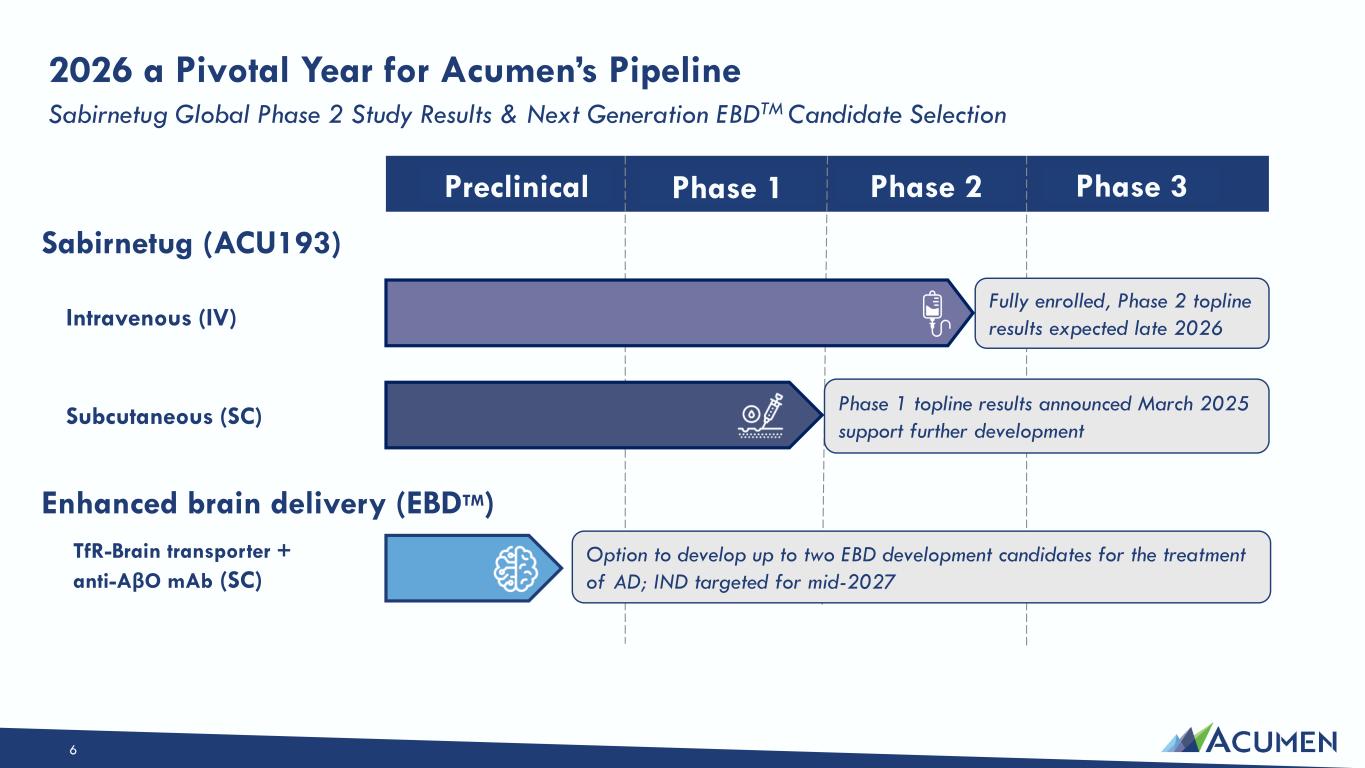
6 Phase 1 Phase 2 Phase 3Preclinical Intravenous (IV) Subcutaneous (SC) Fully enrolled, Phase 2 topline results expected late 2026 Phase 1 topline results announced March 2025 support further development TfR-Brain transporter + anti-AβO mAb (SC) Option to develop up to two EBD development candidates for the treatment of AD; IND targeted for mid-2027 Sabirnetug (ACU193) Enhanced brain delivery (EBDTM) 2026 a Pivotal Year for Acumen’s Pipeline Sabirnetug Global Phase 2 Study Results & Next Generation EBDTM Candidate Selection

Acumen Leadership Team Experienced in AD/Neuro Drug Development Acumen team has decades of experience in Alzheimer’s drug discovery and development AMY SCHACTERLE, PHD Chief Regulatory Officer, Head of Quality PAUL SHUGHRUE, PHD VP, Research & Strategy JASNA JERECIC, PHD Disease Area Strategy Lead ERIC SIEMERS, MD Chief Medical Officer MATT ZUGA Chief Financial Officer & Chief Business Officer RUSSELL BARTON Chief Operating Officer DANIEL O'CONNELL Chief Executive Officer SIEW TIN GAN Head of Clinical Operations 7 JULIE BOCKENSTETTE Chief People Officer JAMES DOHERTY, PHD President & Chief Development Officer LIEAN SCHENK SVP, Head of CMC LAURA ROSEN, MD, PHD SVP, Clinical Development DEREK MEISNER, JD Chief Legal Officer

Milestones Achieved and Upcoming ~$136M Cash, cash equivalents and marketable securities as of Sept. 30, 2025 8 MILESTONES TIMING STATUS Initiation of ALTITUDE-AD Phase 2 trial 2Q2024 ✓ Completion of enrollment of ALTITUDE-AD 1Q2025 ✓ Phase 1 subcutaneous topline results 1Q2025 ✓ EBDTM non-clinical data package Early 2026 ✓ ALTITUDE-AD topline results Late 2026 Acumen expects its cash runway to extend into early 2027

Amyloid Beta Oligomers in AD Sabirnetug (ACU193): monoclonal antibody (mAb) highly selective for toxic AβOs
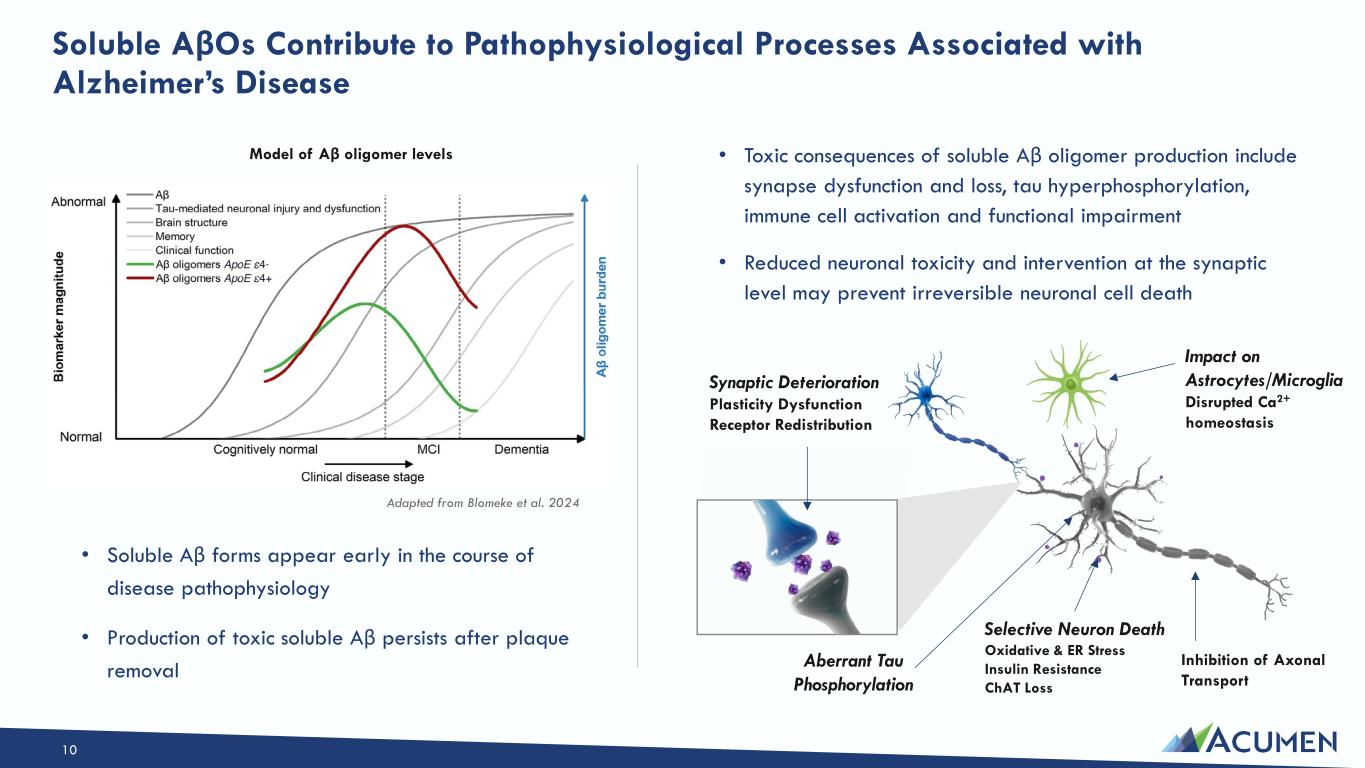
Soluble AβOs Contribute to Pathophysiological Processes Associated with Alzheimer’s Disease • Toxic consequences of soluble Aβ oligomer production include synapse dysfunction and loss, tau hyperphosphorylation, immune cell activation and functional impairment • Reduced neuronal toxicity and intervention at the synaptic level may prevent irreversible neuronal cell death Aberrant Tau Phosphorylation Selective Neuron Death Oxidative & ER Stress Insulin Resistance ChAT Loss Synaptic Deterioration Plasticity Dysfunction Receptor Redistribution Impact on Astrocytes/Microglia Disrupted Ca2+ homeostasis Inhibition of Axonal Transport 10 Adapted from Blomeke et al. 2024 Model of Aβ oligomer levels • Soluble Aβ forms appear early in the course of disease pathophysiology • Production of toxic soluble Aβ persists after plaque removal
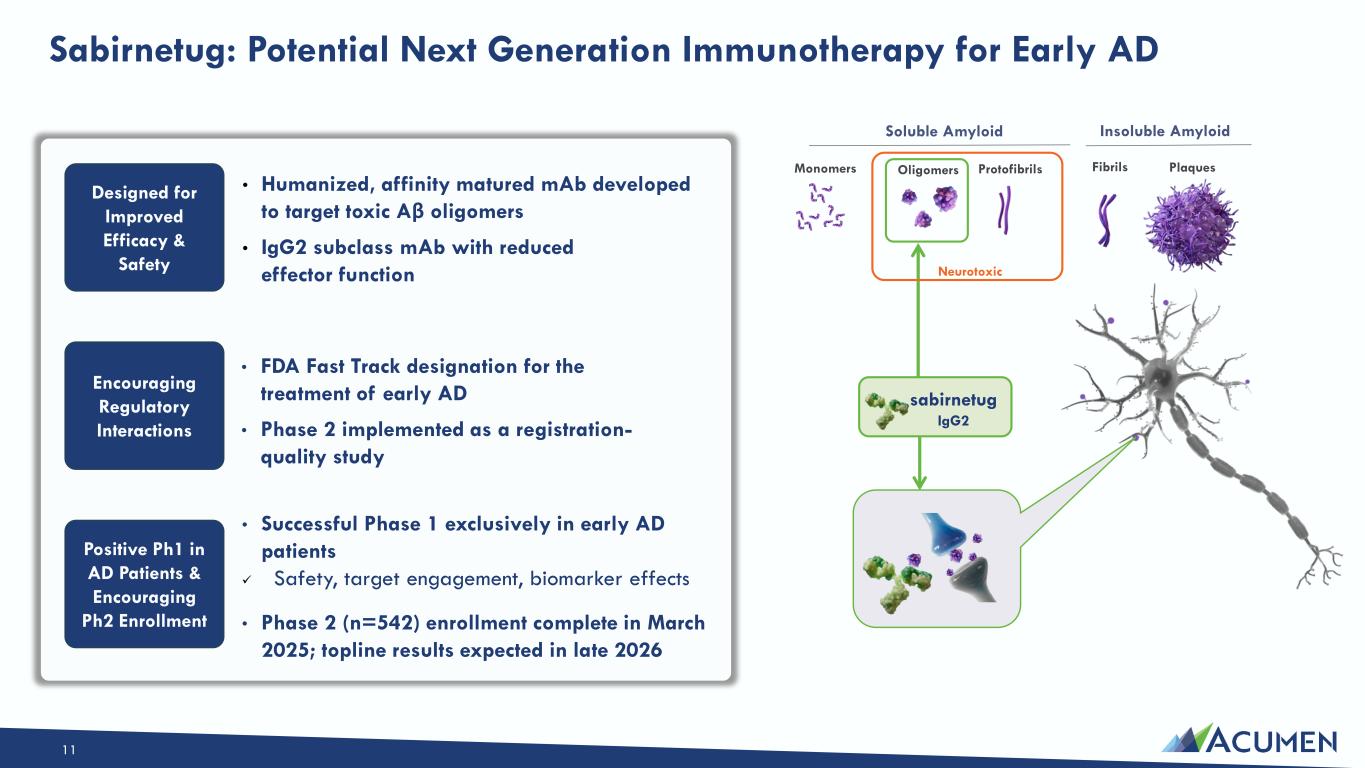
Sabirnetug: Potential Next Generation Immunotherapy for Early AD 11 • Humanized, affinity matured mAb developed to target toxic Aβ oligomers • IgG2 subclass mAb with reduced effector function Designed for Improved Efficacy & Safety Encouraging Regulatory Interactions • FDA Fast Track designation for the treatment of early AD • Phase 2 implemented as a registration- quality study Positive Ph1 in AD Patients & Encouraging Ph2 Enrollment • Successful Phase 1 exclusively in early AD patients ✓ Safety, target engagement, biomarker effects • Phase 2 (n=542) enrollment complete in March 2025; topline results expected in late 2026 Monomers Oligomers Protofibrils Fibrils Plaques Soluble Amyloid Insoluble Amyloid sabirnetug IgG2 Neurotoxic

Sabirnetug
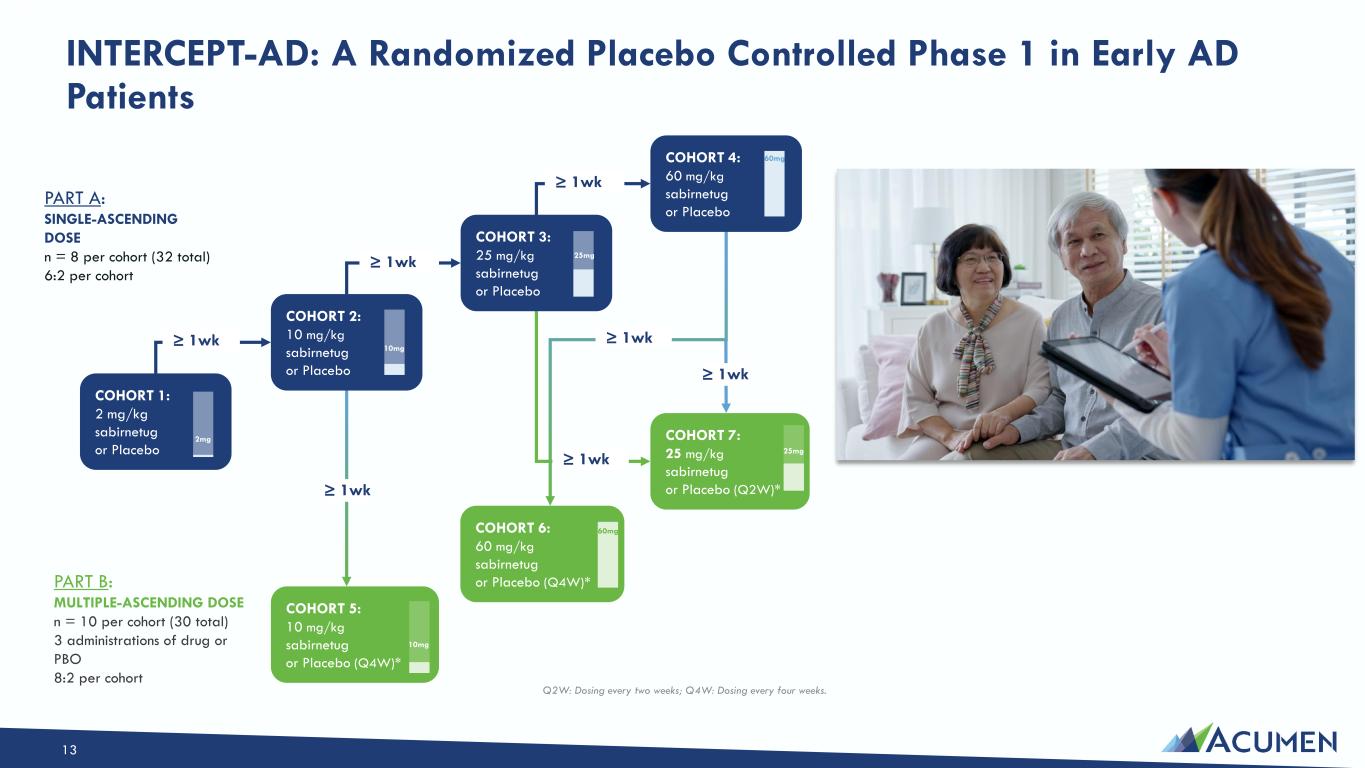
Q2W: Dosing every two weeks; Q4W: Dosing every four weeks. PART A: SINGLE-ASCENDING DOSE n = 8 per cohort (32 total) 6:2 per cohort PART B: MULTIPLE-ASCENDING DOSE n = 10 per cohort (30 total) 3 administrations of drug or PBO 8:2 per cohort COHORT 1: 2 mg/kg sabirnetug or Placebo 2mg COHORT 2: 10 mg/kg sabirnetug or Placebo 10mg COHORT 3: 25 mg/kg sabirnetug or Placebo 25mg COHORT 4: 60 mg/kg sabirnetug or Placebo 60mg COHORT 5: 10 mg/kg sabirnetug or Placebo (Q4W)* 10mg COHORT 6: 60 mg/kg sabirnetug or Placebo (Q4W)* 60mg COHORT 7: 25 mg/kg sabirnetug or Placebo (Q2W)* ≥ 1wk ≥ 1wk ≥ 1wk ≥ 1wk ≥ 1wk ≥ 1wk 25mg ≥ 1wk INTERCEPT-AD: A Randomized Placebo Controlled Phase 1 in Early AD Patients 13
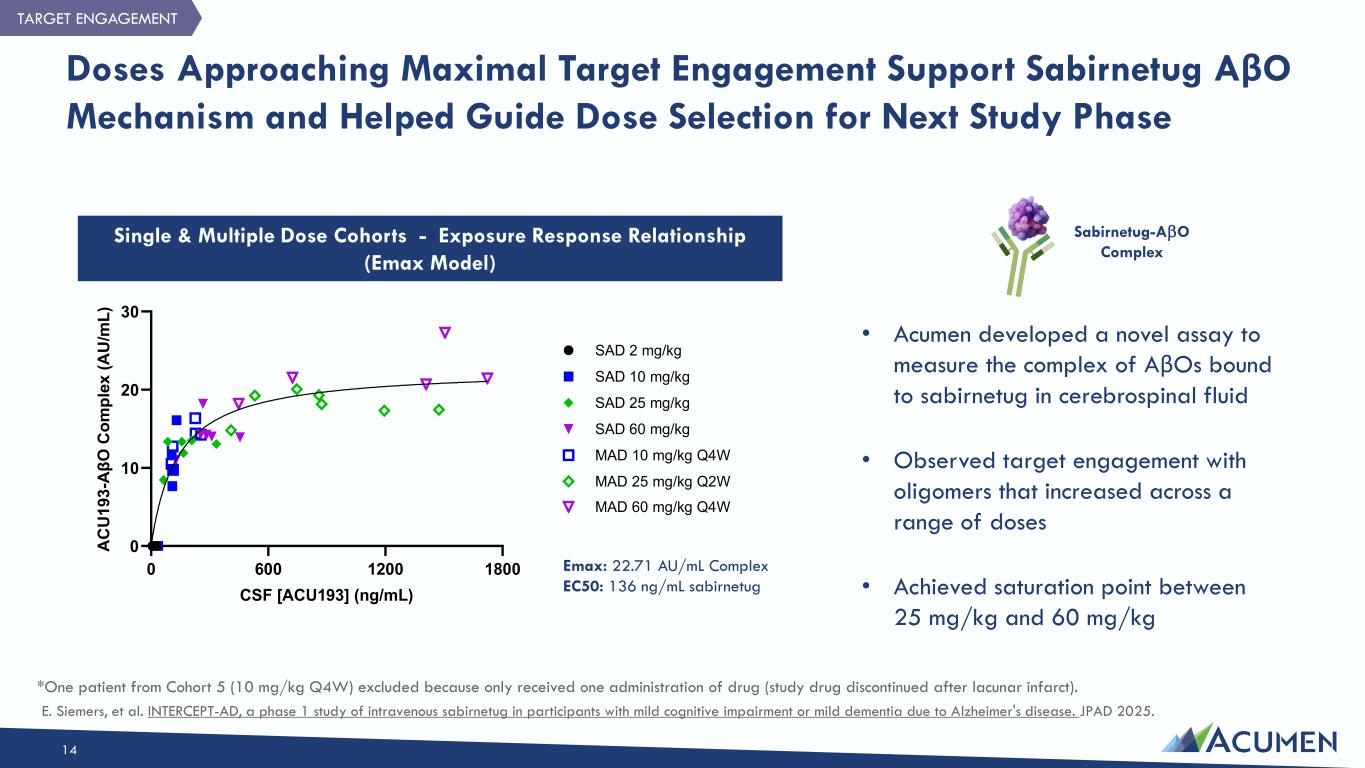
Doses Approaching Maximal Target Engagement Support Sabirnetug AβO Mechanism and Helped Guide Dose Selection for Next Study Phase 14 Single & Multiple Dose Cohorts - Exposure Response Relationship (Emax Model) Emax: 22.71 AU/mL Complex EC50: 136 ng/mL sabirnetug 0 600 1200 1800 0 10 20 30 CSF [ACU193] (ng/mL) A C U 1 9 3 -A β O C o m p le x ( A U /m L ) SAD 2 mg/kg SAD 10 mg/kg SAD 25 mg/kg SAD 60 mg/kg MAD 10 mg/kg Q4W MAD 60 mg/kg Q4W MAD 25 mg/kg Q2W Sabirnetug-AO Complex TARGET ENGAGEMENT *One patient from Cohort 5 (10 mg/kg Q4W) excluded because only received one administration of drug (study drug discontinued after lacunar infarct). E. Siemers, et al. INTERCEPT-AD, a phase 1 study of intravenous sabirnetug in participants with mild cognitive impairment or mild dementia due to Alzheimer's disease. JPAD 2025. • Acumen developed a novel assay to measure the complex of AβOs bound to sabirnetug in cerebrospinal fluid • Observed target engagement with oligomers that increased across a range of doses • Achieved saturation point between 25 mg/kg and 60 mg/kg
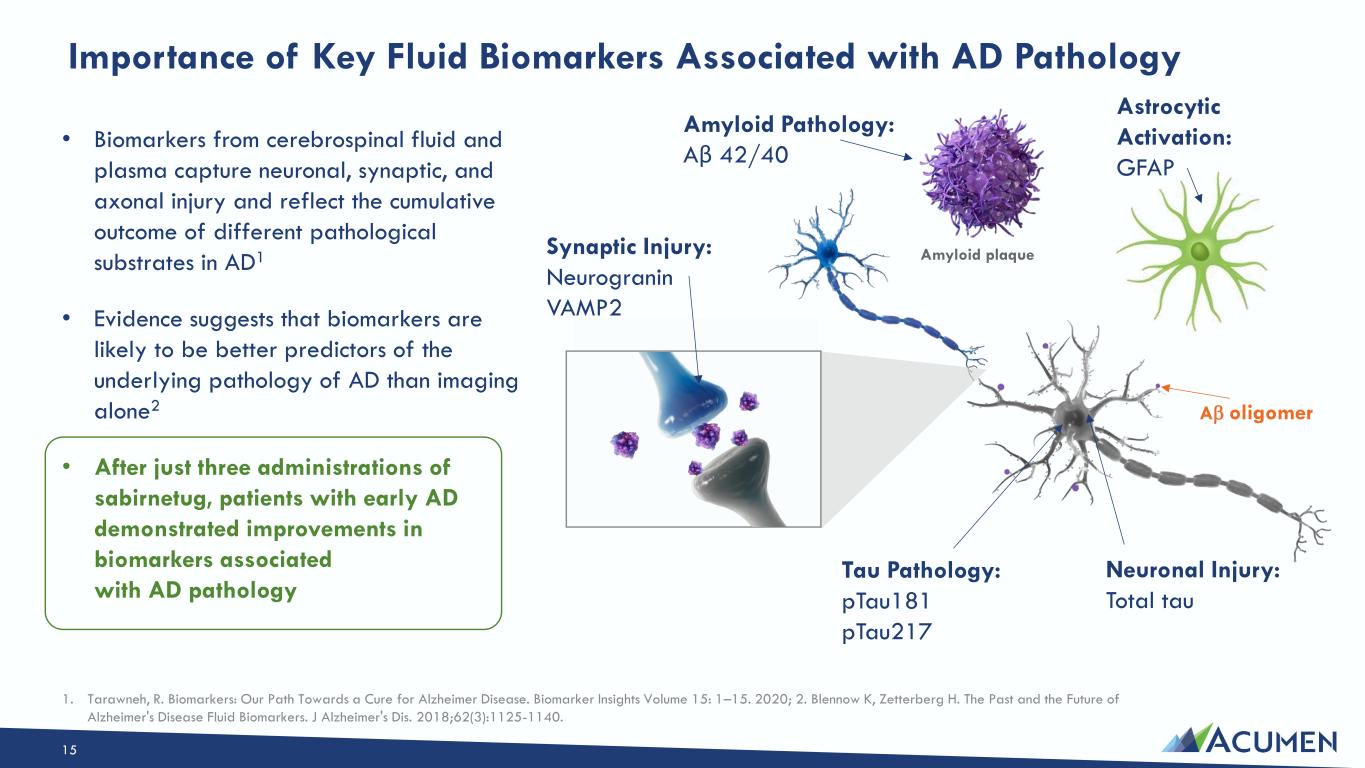
Importance of Key Fluid Biomarkers Associated with AD Pathology 15 Tau Pathology: pTau181 pTau217 Neuronal Injury: Total tau Synaptic Injury: Neurogranin VAMP2 Amyloid Pathology: Aβ 42/40 Astrocytic Activation: GFAP • Biomarkers from cerebrospinal fluid and plasma capture neuronal, synaptic, and axonal injury and reflect the cumulative outcome of different pathological substrates in AD1 • Evidence suggests that biomarkers are likely to be better predictors of the underlying pathology of AD than imaging alone2 • After just three administrations of sabirnetug, patients with early AD demonstrated improvements in biomarkers associated with AD pathology 1. Tarawneh, R. Biomarkers: Our Path Towards a Cure for Alzheimer Disease. Biomarker Insights Volume 15: 1–15. 2020; 2. Blennow K, Zetterberg H. The Past and the Future of Alzheimer's Disease Fluid Biomarkers. J Alzheimer's Dis. 2018;62(3):1125-1140. A oligomer Amyloid plaque
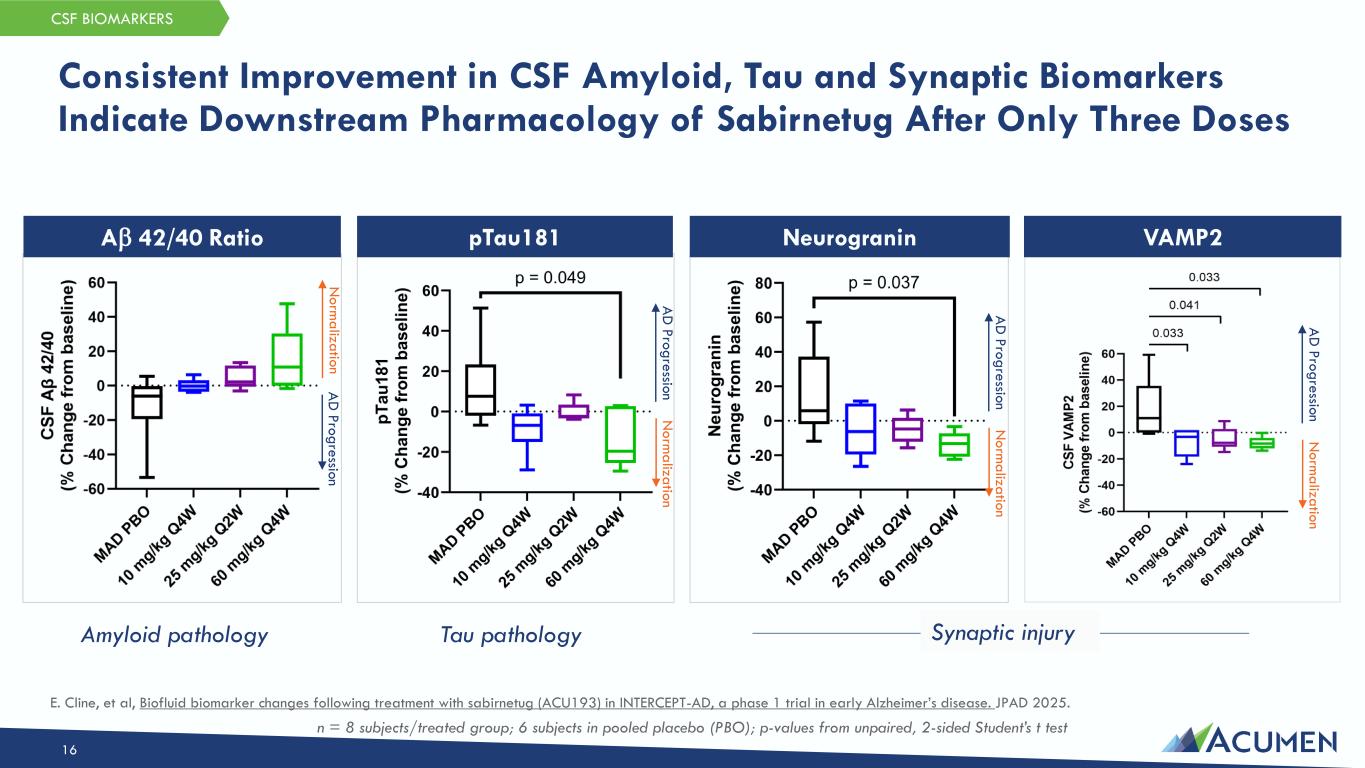
Consistent Improvement in CSF Amyloid, Tau and Synaptic Biomarkers Indicate Downstream Pharmacology of Sabirnetug After Only Three Doses 16 CSF BIOMARKERS pTau181 NeurograninA 42/40 Ratio VAMP2 Tau pathologyAmyloid pathology Synaptic injury n = 8 subjects/treated group; 6 subjects in pooled placebo (PBO); p-values from unpaired, 2-sided Student’s t test N o rm a liza tio n A D P ro g re ssio n N o rm a liza tio n A D P ro g re ssio n N o rm a liza tio n A D P ro g re ssio n N o rm a liza tio n A D P ro g re ssio n E. Cline, et al, Biofluid biomarker changes following treatment with sabirnetug (ACU193) in INTERCEPT-AD, a phase 1 trial in early Alzheimer’s disease. JPAD 2025.
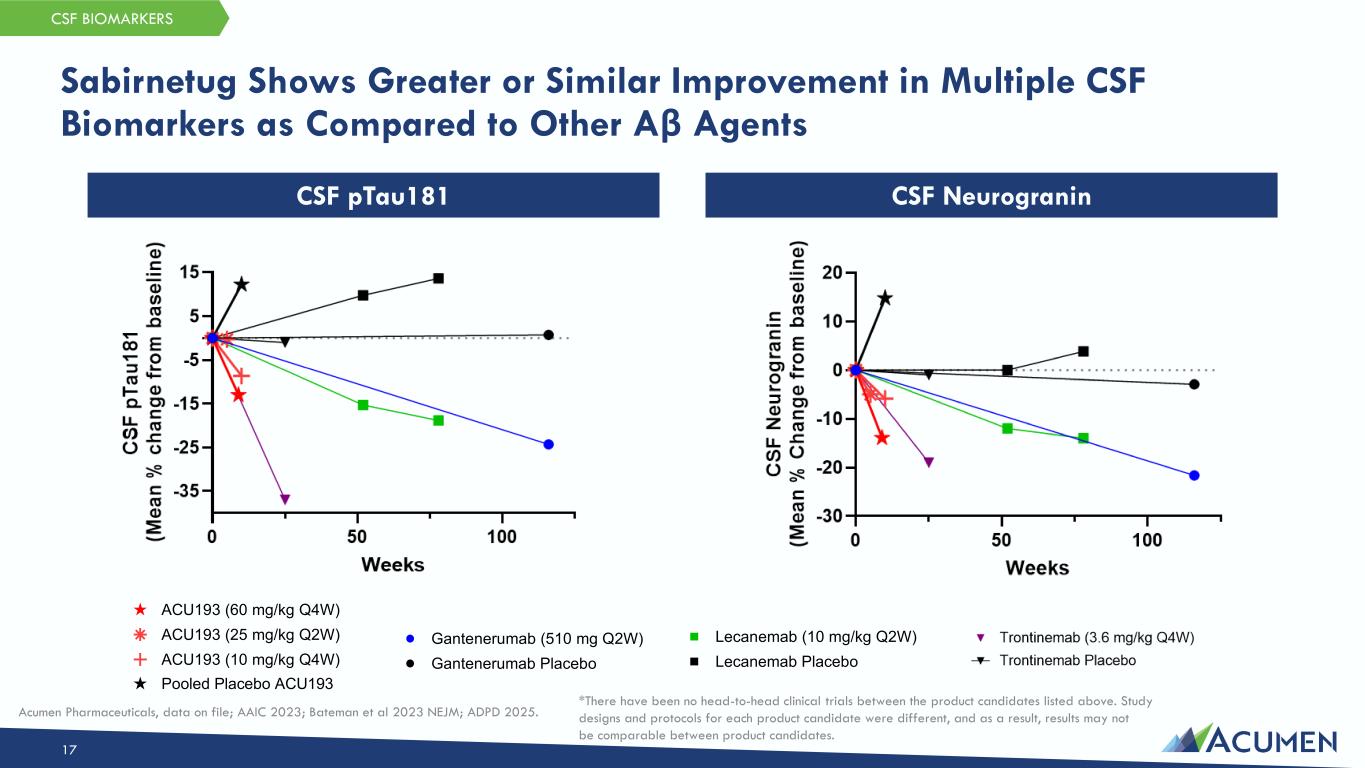
Sabirnetug Shows Greater or Similar Improvement in Multiple CSF Biomarkers as Compared to Other Aβ Agents 17 CSF BIOMARKERS CSF NeurograninCSF pTau181 50 100 -35 -25 -15 -5 5 15 Weeks C S F p T a u 1 8 1 (M e a n % c h a n g e f ro m b a s e li n e ) Gantenerumab (510 mg Q2W) Gantenerumab Placebo ACU193 (60 mg/kg Q4W) ACU193 (25 mg/kg Q2W) ACU193 (10 mg/kg Q4W) Pooled Placebo ACU193 Lecanemab (10 mg/kg Q2W) Lecanemab Placebo Trontinemab (3.6 mg/kg Q4W) C S F p T a u 1 8 1 (M e a n % c h a n g e f ro m b a s e li n e ) 50 100 -35 -25 -15 -5 5 15 Weeks C S F p T a u 1 8 1 (M e a n % c h a n g e f ro m b a s e li n e ) Gantenerumab (510 mg Q2W) Gantenerumab Placebo ACU193 (60 mg/kg Q4W) ACU193 (25 mg/kg Q2W) ACU193 (10 mg/kg Q4W) Pooled Placebo ACU193 Lecanemab (10 mg/kg Q2W) Lecanemab Placebo Trontinemab (3.6 mg/kg Q4W) 50 100 -35 -25 -15 -5 5 15 Weeks C S F p T a u 1 8 1 (M e a n % c h a n g e f ro m b a s e li n e ) Gantenerumab (510 mg Q2W) Gantenerumab Placebo ACU193 (60 mg/kg Q4W) ACU193 (25 mg/kg Q2W) ACU193 (10 mg/kg Q4W) Pooled Placebo ACU193 Lecanemab (10 mg/kg Q2W) Lecanemab Placebo Trontinemab (3.6 mg/kg Q4W) *There have been no head-to-head clinical trials between the product candidates listed above. Study designs and protocols for each product candidate were different, and as a result, results may not be comparable between product candidates. Acumen Pharmaceuticals, data on file; AAIC 2023; Bateman et al 2023 NEJM; ADPD 2025.
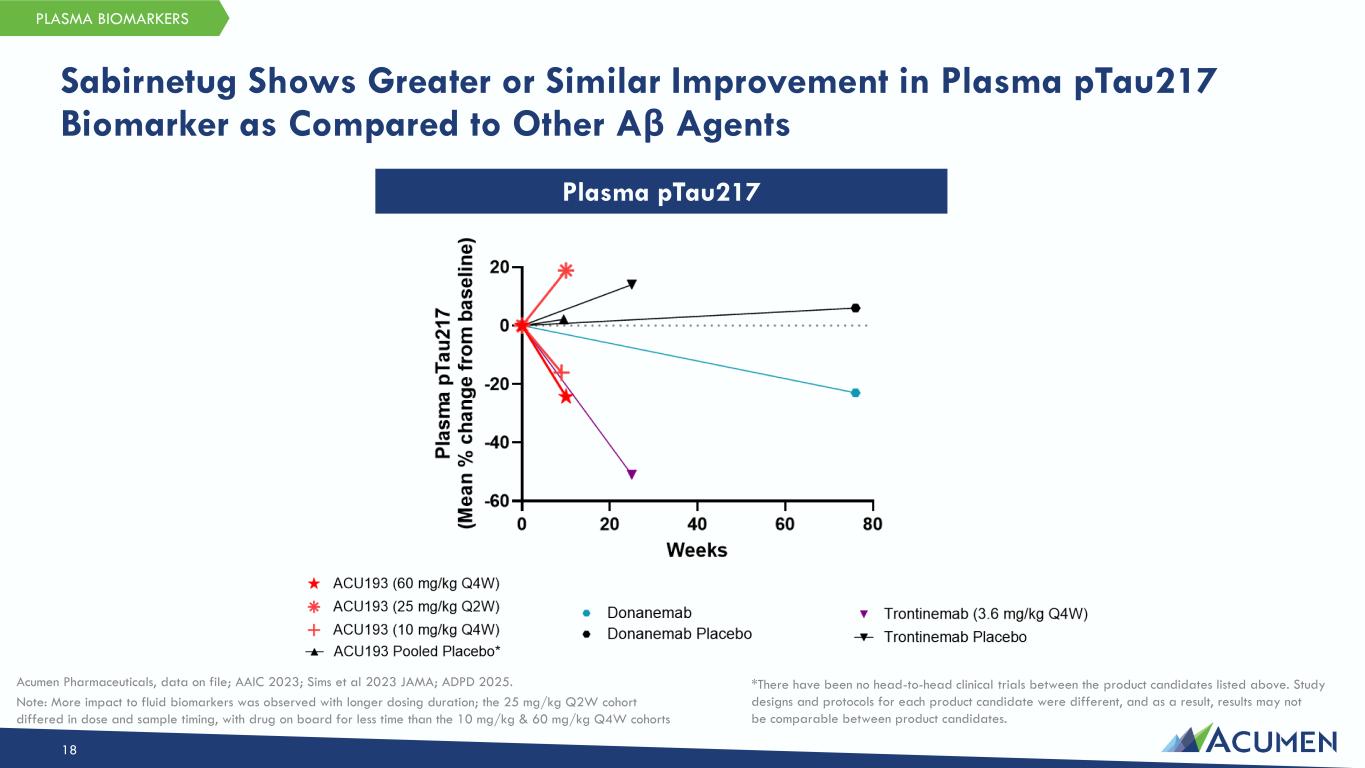
Sabirnetug Shows Greater or Similar Improvement in Plasma pTau217 Biomarker as Compared to Other Aβ Agents 18 PLASMA BIOMARKERS Plasma pTau217 *There have been no head-to-head clinical trials between the product candidates listed above. Study designs and protocols for each product candidate were different, and as a result, results may not be comparable between product candidates. Acumen Pharmaceuticals, data on file; AAIC 2023; Sims et al 2023 JAMA; ADPD 2025. Note: More impact to fluid biomarkers was observed with longer dosing duration; the 25 mg/kg Q2W cohort differed in dose and sample timing, with drug on board for less time than the 10 mg/kg & 60 mg/kg Q4W cohorts
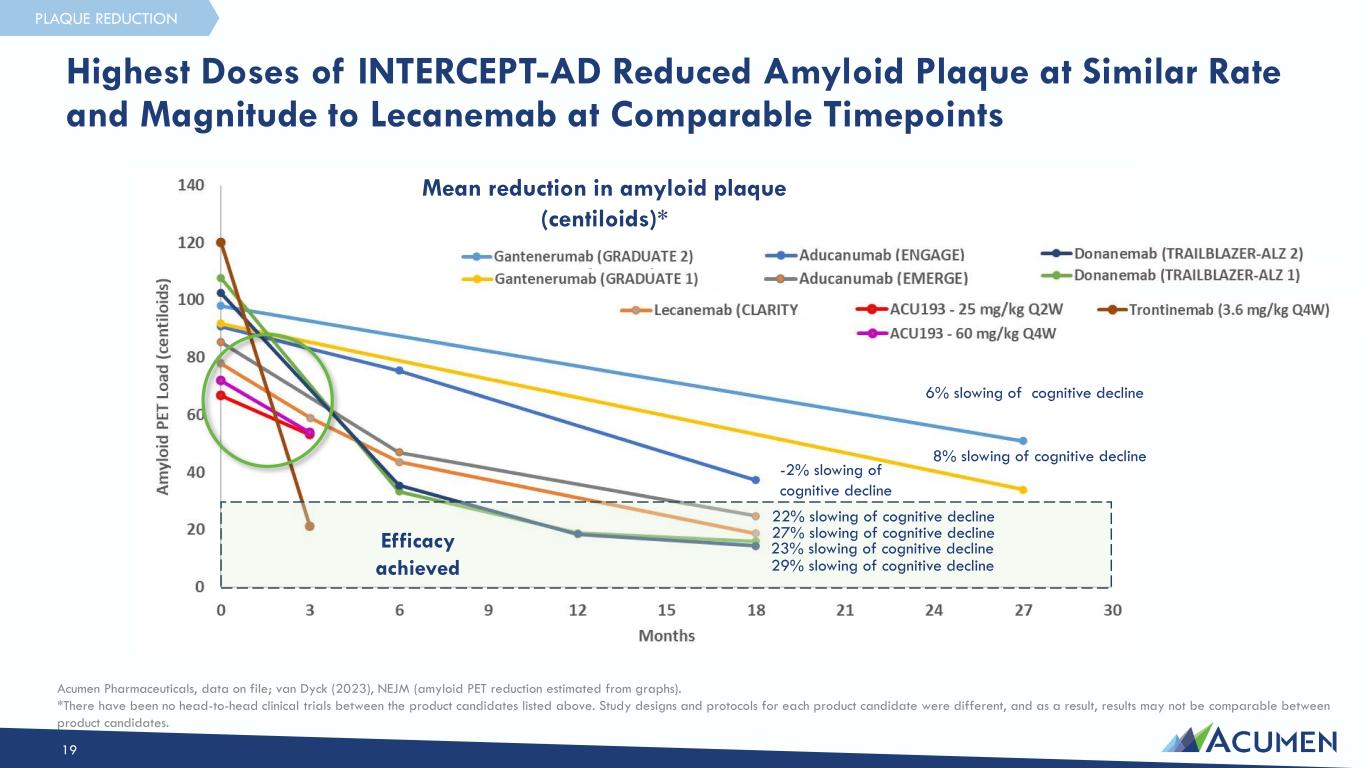
Highest Doses of INTERCEPT-AD Reduced Amyloid Plaque at Similar Rate and Magnitude to Lecanemab at Comparable Timepoints 6% slowing of cognitive decline 8% slowing of cognitive decline -2% slowing of cognitive decline 22% slowing of cognitive decline 27% slowing of cognitive decline 23% slowing of cognitive decline 29% slowing of cognitive decline Efficacy achieved Mean reduction in amyloid plaque (centiloids)* Acumen Pharmaceuticals, data on file; van Dyck (2023), NEJM (amyloid PET reduction estimated from graphs). *There have been no head-to-head clinical trials between the product candidates listed above. Study designs and protocols for each product candidate were different, and as a result, results may not be comparable between product candidates. 19 PLAQUE REDUCTION

Sabirnetug Demonstrates Potential for Best-in-Class Safety Profile Compelling Overall Safety Profile, with Low Incidence of ARIA-E in the INTERCEPT-AD study 20 SAFETY ✓ Limited incidence of ARIA-E • 10 mg/kg Q4W: 1 asymptomatic case • 25 mg/kg Q2W: 1 asymptomatic case • 60 mg/kg Q4W: 2 asymptomatic cases; 1 symptomatic case ✓ No ARIA-E observed in ApoE4 homozygotes (n=6), despite comprising 13% of study • Differentiated from other antibodies that have ARIA-E rates ~30% to ~40% in participants who are E4-homozygotes ✓ Broad therapeutic index with convenient monthly dosing • Safety profile may support attractive benefit/risk option for large portion of patients 5 0 Total ARIA-E cases, or ~10% Cases of ARIA-E in ApoE4 homozygotes N=6 0 Deaths, SAEs Related to Study Drug INTERCEPT-AD Phase 1 Safety Data E. Siemers, et al. INTERCEPT-AD, a phase 1 study of intravenous sabirnetug in participants with mild cognitive impairment or mild dementia due to Alzheimer's disease. JPAD 2025. ApoE4: ε4 allele of Apolipoprotein E

Potential for Differentiated Efficacy Potential for Differentiated Safety INTERCEPT-AD Phase 1 Data Support Potential for Sabirnetug to Offer Next Generation Efficacy and Safety 21 ✓ Compelling safety profile with low incidence of ARIA-E ✓ Absence of ARIA-E observed in ApoE4 homozygotes ✓ Broad potential therapeutic index with convenient monthly dosing ✓ First mAb to demonstrate selective target engagement of AβOs (most toxic form of Aβ) ✓ Rapid, significant plaque reduction comparable to the current market front-runners at similar timepoints ✓ Improvement of AD biomarkers in CSF and plasma are a strong indication of downstream effects Key Takeaways from INTERCEPT-AD

R a n d o m iz a ti o n 1 :1 :1 Sabirnetug 35mg/kg Q4W (n ~180) Sabirnetug 50mg/kg Q4W* (n ~180) Placebo Q4W (n ~180) Primary Endpoint Change in iADRS1 at 18 months Secondary Endpoints1 CDR-SB, ADAS- Cog13, ADCS-ADL, AD biomarkers 12-month open label extension Objective: To evaluate the clinical efficacy, safety and tolerability of sabirnetug Patient population: Patients with early AD (MCI or mild dementia due to early AD) ALTITUDE-AD Phase 2 Study Results Expected in Late 2026 1. iADRS: Integrated Alzheimer's Disease Rating Scale; CDR-SB: Clinical Dementia Rating – Sum of Boxes; ADAS-cog: Alzheimer's Disease Assessment Scale – Cognitive Subscale; ADCS-ADL: Alzheimer's Disease Cooperative Study – Activities of Daily Living 22 *Titration of sabirnetug 35mg/kg Q4W for two doses. • 542-participant study fully enrolled in March 2025 (U.S., Canada, U.K., Germany, Spain) • Open label extension portion of ALTITUDE-AD initiated in November 2025 • Topline results expected in late 2026 Key Highlights
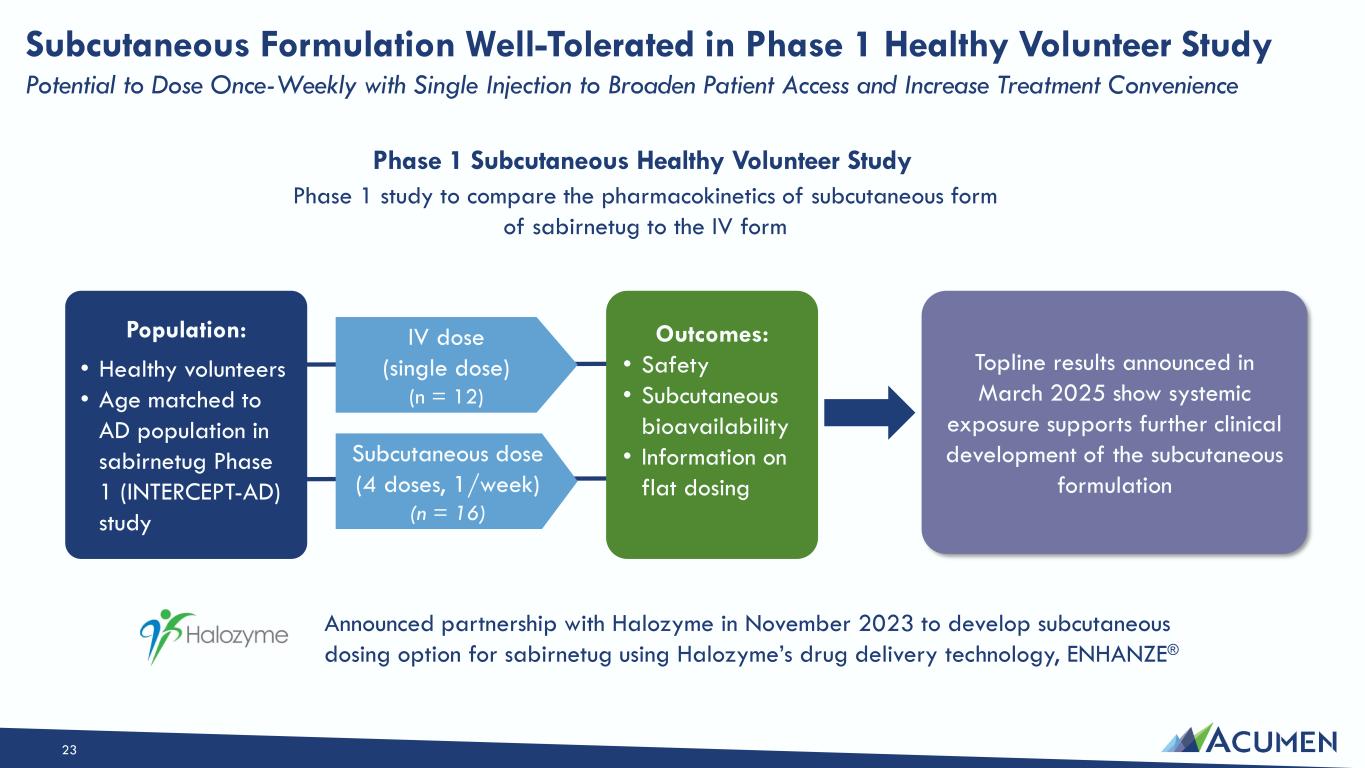
Subcutaneous Formulation Well-Tolerated in Phase 1 Healthy Volunteer Study Potential to Dose Once-Weekly with Single Injection to Broaden Patient Access and Increase Treatment Convenience Topline results announced in March 2025 show systemic exposure supports further clinical development of the subcutaneous formulation 23 Phase 1 study to compare the pharmacokinetics of subcutaneous form of sabirnetug to the IV form Outcomes: • Safety • Subcutaneous bioavailability • Information on flat dosing Population: • Healthy volunteers • Age matched to AD population in sabirnetug Phase 1 (INTERCEPT-AD) study IV dose (single dose) (n = 12) Subcutaneous dose (4 doses, 1/week) (n = 16) Phase 1 Subcutaneous Healthy Volunteer Study Announced partnership with Halozyme in November 2023 to develop subcutaneous dosing option for sabirnetug using Halozyme’s drug delivery technology, ENHANZE®

Sabirnetug IP & Market Exclusivity • Exclusive, perpetual, irrevocable, worldwide, royalty-free license from Merck to its Amyloid Derived Diffusible Ligand (ADDL) IP including issued sabirnetug patents • Sabirnetug Global IP estate: ✓ Issued patents in 19 countries ✓ Composition of matter patents and methods of use run into July 2031 ✓ Patent term extensions may be available, 3-5 years depending on jurisdiction • Biologics market exclusivity is expected for sabirnetug as a novel biologic drug ✓ US provides 12 years market exclusivity for novel biologics ✓ Europe provides 10 years of market exclusivity for novel biologics • EBDTM program includes Acumen wholly-owned AO-selective mAbs expected to generate novel IP including Composition of Matter Patent(s) 24

Enhanced Brain Delivery (EBDTM)
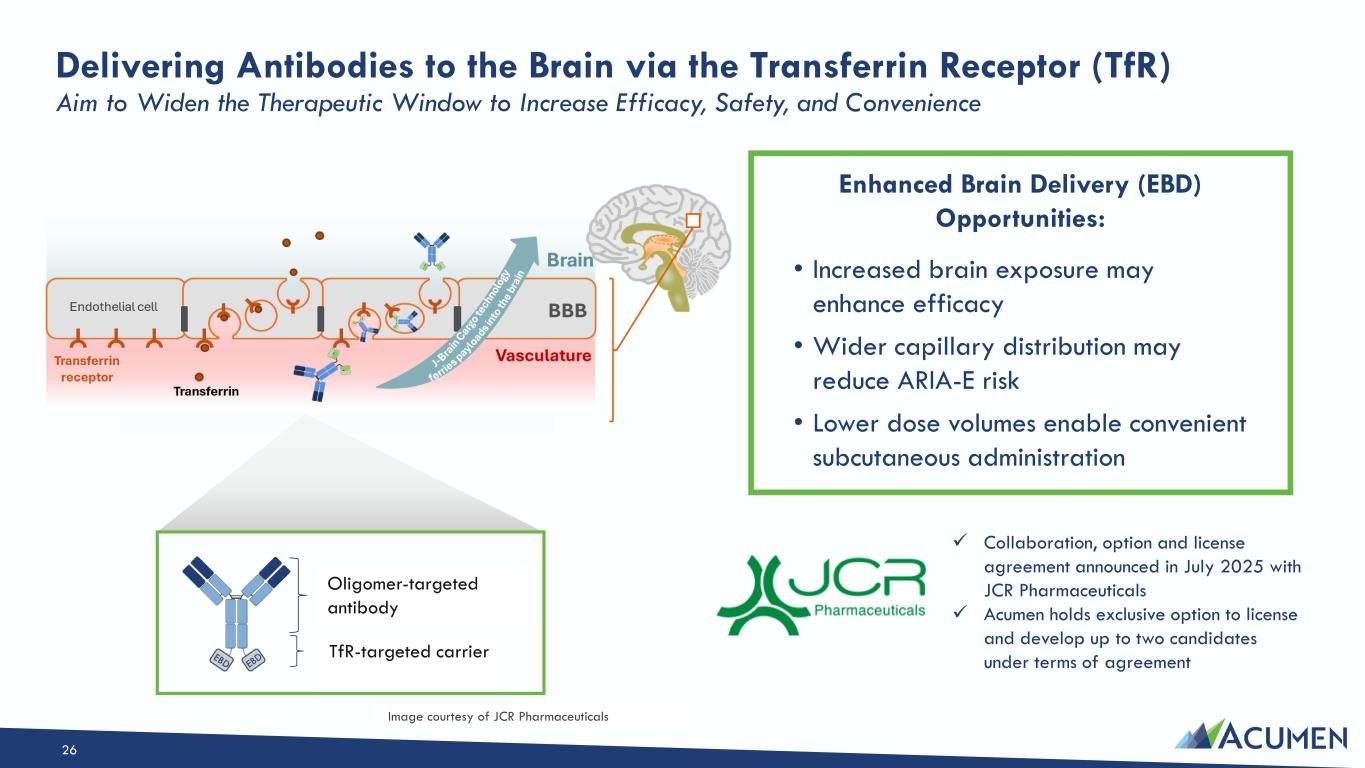
26 Delivering Antibodies to the Brain via the Transferrin Receptor (TfR) Aim to Widen the Therapeutic Window to Increase Efficacy, Safety, and Convenience • Increased brain exposure may enhance efficacy • Wider capillary distribution may reduce ARIA-E risk • Lower dose volumes enable convenient subcutaneous administration Enhanced Brain Delivery (EBD) Opportunities: Oligomer-targeted antibody TfR-targeted carrier ✓ Collaboration, option and license agreement announced in July 2025 with JCR Pharmaceuticals ✓ Acumen holds exclusive option to license and develop up to two candidates under terms of agreement Image courtesy of JCR Pharmaceuticals

Acumen's EBD May Improve Efficacy, Safety and Delivery Compared to Non-AβO Selective Antibodies Differentiated Cargo (AβO-selective mAb) 27 Differentiated Aβ targeting Validated TfR-binding Validated BBB Carrier Technology (TfR-targeting) Acumen EBD Candidates • Targets synaptotoxic A species • Robust fluid biomarker results in Phase 1b INTERCEPT-AD trial • Demonstrated low ARIA-E overall in Phase 1b, including no ARIA- E observed in APOE 4/4 carriers • Low rate of infusion related reactions observed to date clinically • Candidate mAbs: ACU193 and ACU234 • Enhance brain penetration versus native antibody to enable low- volume SC delivery • Potential for lower ARIA rate due to TfR prevalence on capillary bed • Little to no anemia observed with JCR technologies = Enhanced efficacy: Leverage improved brain delivery/ distribution and Aβ oligomer targeting Best-in-class safety: Lower ARIA, anemia, IRRs Designed for SC administration: Low dose, formulation, stability ApoE4: ε4 allele of Apolipoprotein E
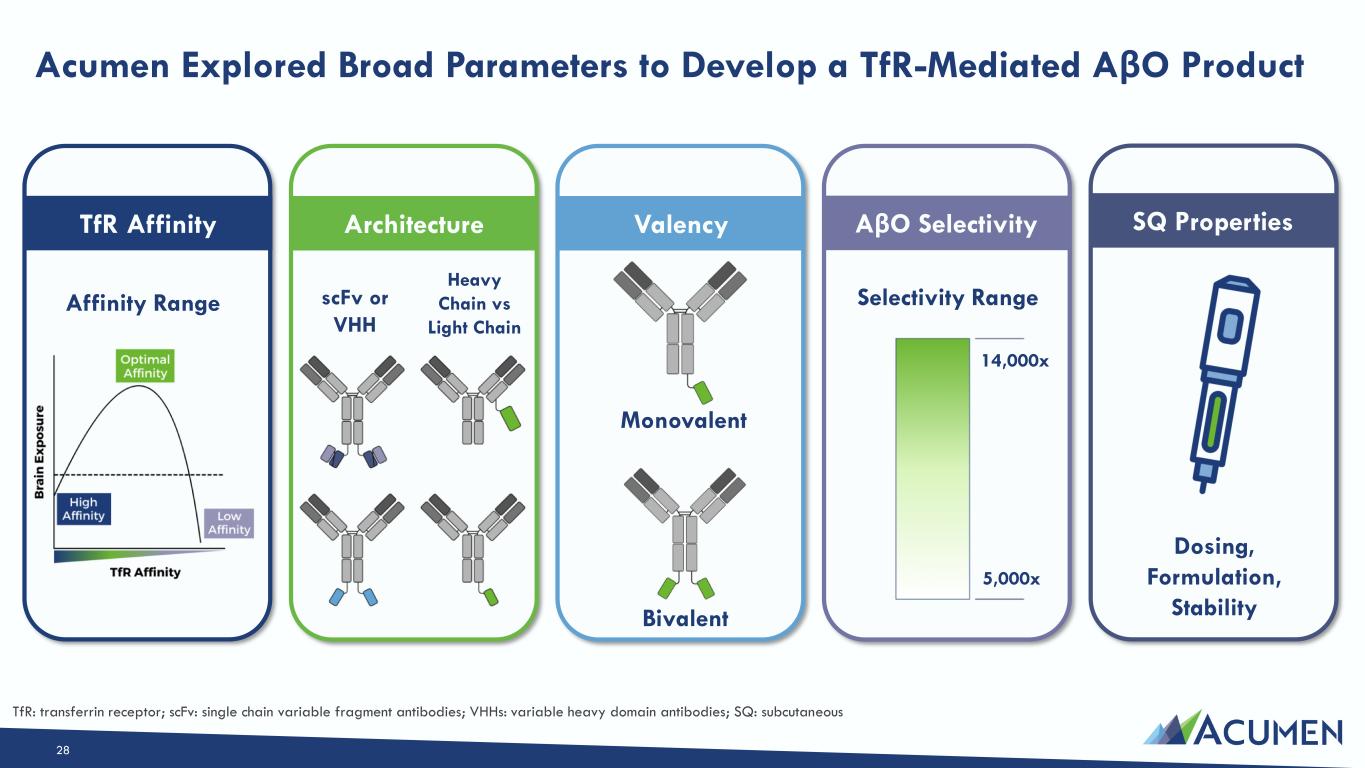
Acumen Explored Broad Parameters to Develop a TfR-Mediated AβO Product 28 TfR Affinity Architecture Valency AβO Selectivity scFv or VHH Heavy Chain vs Light Chain Monovalent Bivalent Affinity Range Selectivity Range SQ Properties 14,000x 5,000x Dosing, Formulation, Stability TfR: transferrin receptor; scFv: single chain variable fragment antibodies; VHHs: variable heavy domain antibodies; SQ: subcutaneous
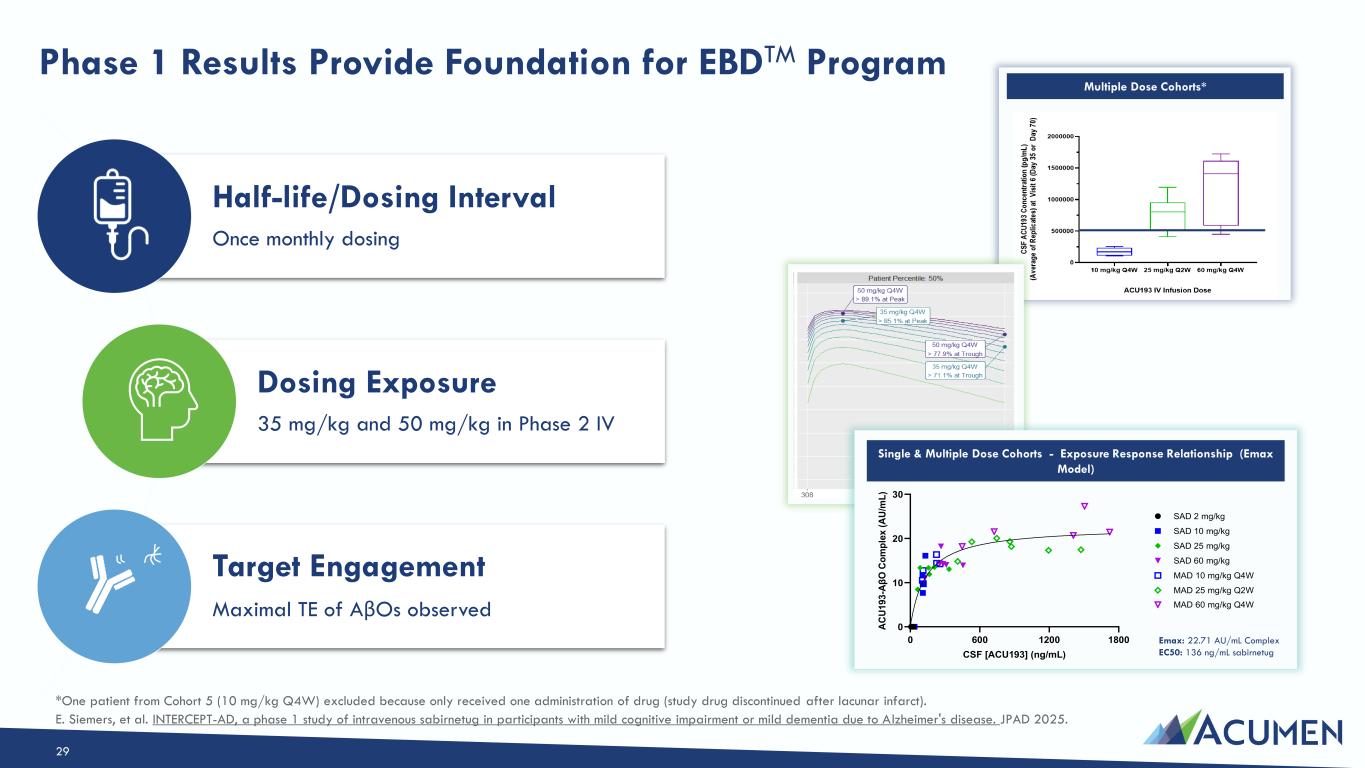
Multiple Dose Cohorts* Half-life/Dosing Interval Once monthly dosing Dosing Exposure 35 mg/kg and 50 mg/kg in Phase 2 IV Target Engagement Maximal TE of AβOs observed Phase 1 Results Provide Foundation for EBDTM Program 29 Single & Multiple Dose Cohorts - Exposure Response Relationship (Emax Model) Emax: 22.71 AU/mL Complex EC50: 136 ng/mL sabirnetug 0 600 1200 1800 0 10 20 30 CSF [ACU193] (ng/mL) A C U 1 9 3 -A β O C o m p le x ( A U /m L ) SAD 2 mg/kg SAD 10 mg/kg SAD 25 mg/kg SAD 60 mg/kg MAD 10 mg/kg Q4W MAD 60 mg/kg Q4W MAD 25 mg/kg Q2W *One patient from Cohort 5 (10 mg/kg Q4W) excluded because only received one administration of drug (study drug discontinued after lacunar infarct). E. Siemers, et al. INTERCEPT-AD, a phase 1 study of intravenous sabirnetug in participants with mild cognitive impairment or mild dementia due to Alzheimer's disease. JPAD 2025.

Mouse Surrogate Antibodies Developed to Explore EBDTM and Target Engagement in a Mouse Model of Alzheimer’s Disease 30 The fusion of anti-oligomer antibodies and J-Brain Cargo (anti-TfR scFv) did not alter the ability of constructs to bind TfR or alter the preferential binding to AβOs Cline et al., CTAD, 2025 234 0 nM Oligomer Binding Monomer Binding 2.182 nM 3.761 M ACU193 0.33 nM 5.34 nM 3.52 nM 0.595 nM 0.448 nM 2.475 nM 6.914 M 5.871 M 8.6 M mTfR: murine transferrin receptor; mscFv: murine single chain variable fragment antibodies 0.319 * *Cline et al., AAIC, 2025
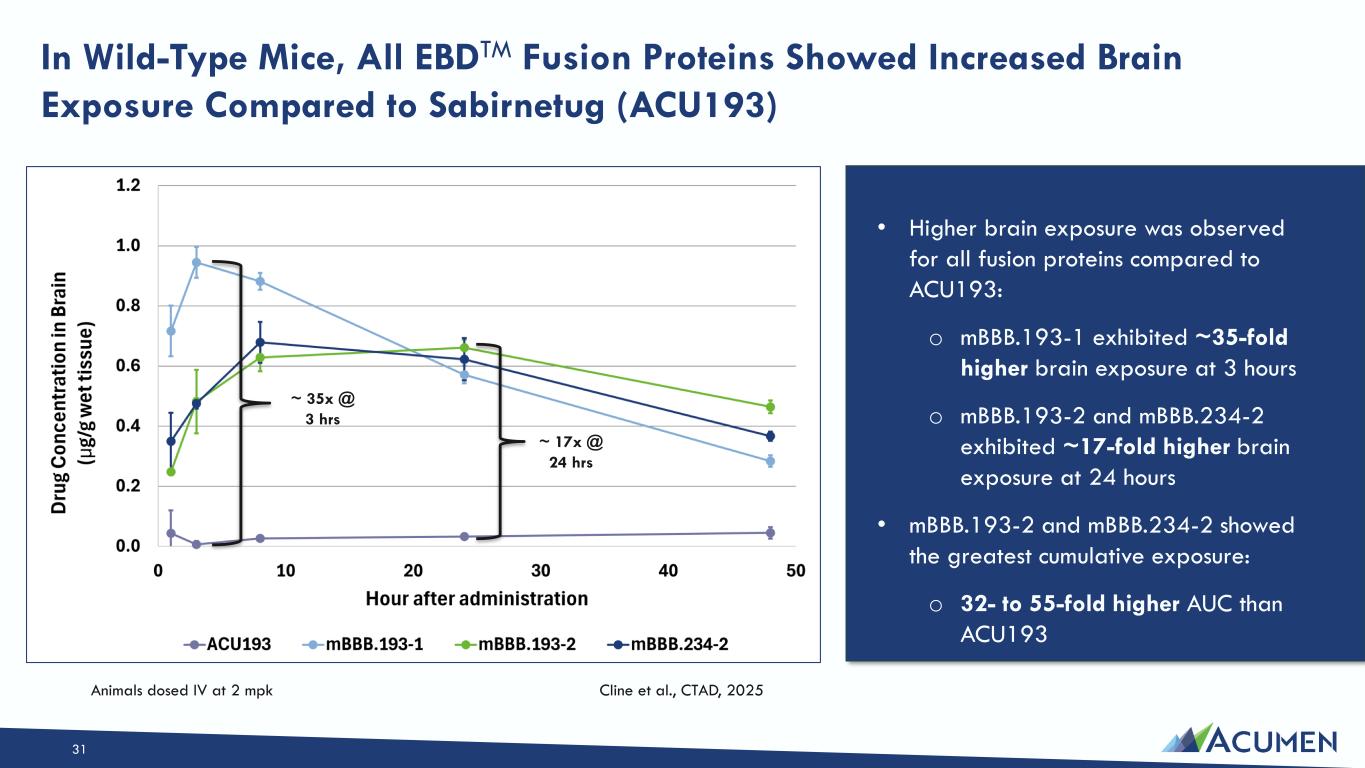
Animals dosed IV at 2 mpk Cline et al., CTAD, 2025 • Higher brain exposure was observed for all fusion proteins compared to ACU193: o mBBB.193-1 exhibited ~35-fold higher brain exposure at 3 hours o mBBB.193-2 and mBBB.234-2 exhibited ~17-fold higher brain exposure at 24 hours • mBBB.193-2 and mBBB.234-2 showed the greatest cumulative exposure: o 32- to 55-fold higher AUC than ACU193 In Wild-Type Mice, All EBDTM Fusion Proteins Showed Increased Brain Exposure Compared to Sabirnetug (ACU193) ~ 35x @ 3 hrs ~ 17x @ 24 hrs 31
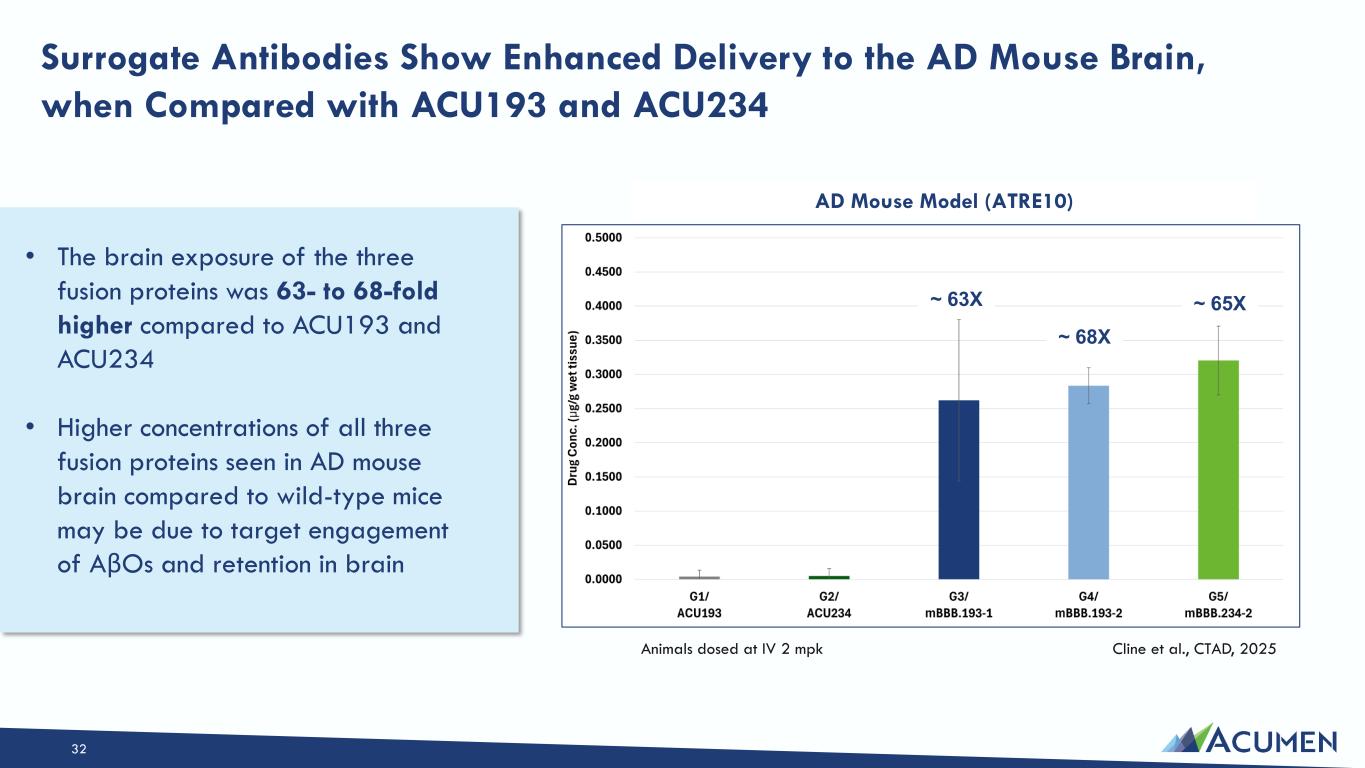
Surrogate Antibodies Show Enhanced Delivery to the AD Mouse Brain, when Compared with ACU193 and ACU234 AD Mouse Model (ATRE10) Animals dosed at IV 2 mpk Cline et al., CTAD, 2025 ~ 63X ~ 68X ~ 65X 32 • The brain exposure of the three fusion proteins was 63- to 68-fold higher compared to ACU193 and ACU234 • Higher concentrations of all three fusion proteins seen in AD mouse brain compared to wild-type mice may be due to target engagement of AβOs and retention in brain

Robust Data Package Supporting EBDTM Construct Selection Inclusive of NHP Primate Data Anemia Risk Brain Distribution Stability & Manufacturability Pharmacokinetics TfR Affinity 33 Aβ Oligomer Binding

Key Takeaways from NHP Study Results 34 ✓ Enhanced Brain Penetration EBD candidates achieved 14-40x higher brain levels in non-human primates compared to native antibodies 24 hours after dosing Lead clinical candidate IND targeted for 2027 ✓ Subcutaneous Dosing Capability Favorable stability profile and enhanced brain delivery support a path to subcutaneous administration with low-volume devices ✓ Low Anemia Risk • Hematology data in non-human primates indicate low potential for anemia: At 24 hours after SC dosing, EBD candidates demonstrated no observed change in red blood cell count, hematocrit, hemoglobin or reticulocyte count • No adverse events observed

✓ Significant and growing Alzheimer’s population in need of additional treatment options ✓ Synaptotoxic AβOs appear early in Alzheimer’s Disease and contribute to its pathophysiological processes; sabirnetug demonstrates high selectivity for AβOs in AD patients ✓ Positive Phase 1 data strengthen potential for sabirnetug to offer next-generation efficacy and safety ✓ Significant interest in ALTITUDE-AD, a Phase 2 study investigating sabirnetug, evidenced by rapid enrollment ✓ Enhanced Brain DeliveryTM program augments portfolio optionality, leveraging Acumen capabilities and assets Acumen Positioned to Deliver Potential Next-Gen Treatment for Early AD 35 Key Takeaways Next Steps ✓ EBDTM program pre-clinical candidate (PCC) data announced in early 2026 ❑ Topline results from ALTITUDE-AD Phase 2 IV study evaluating the clinical efficacy and safety of sabirnetug in patients with MCI or mild dementia due to Alzheimer’s expected in late 2026 ❑ IND for EBD program targeted for mid-2027

Appendix www.acumenpharm.com 36
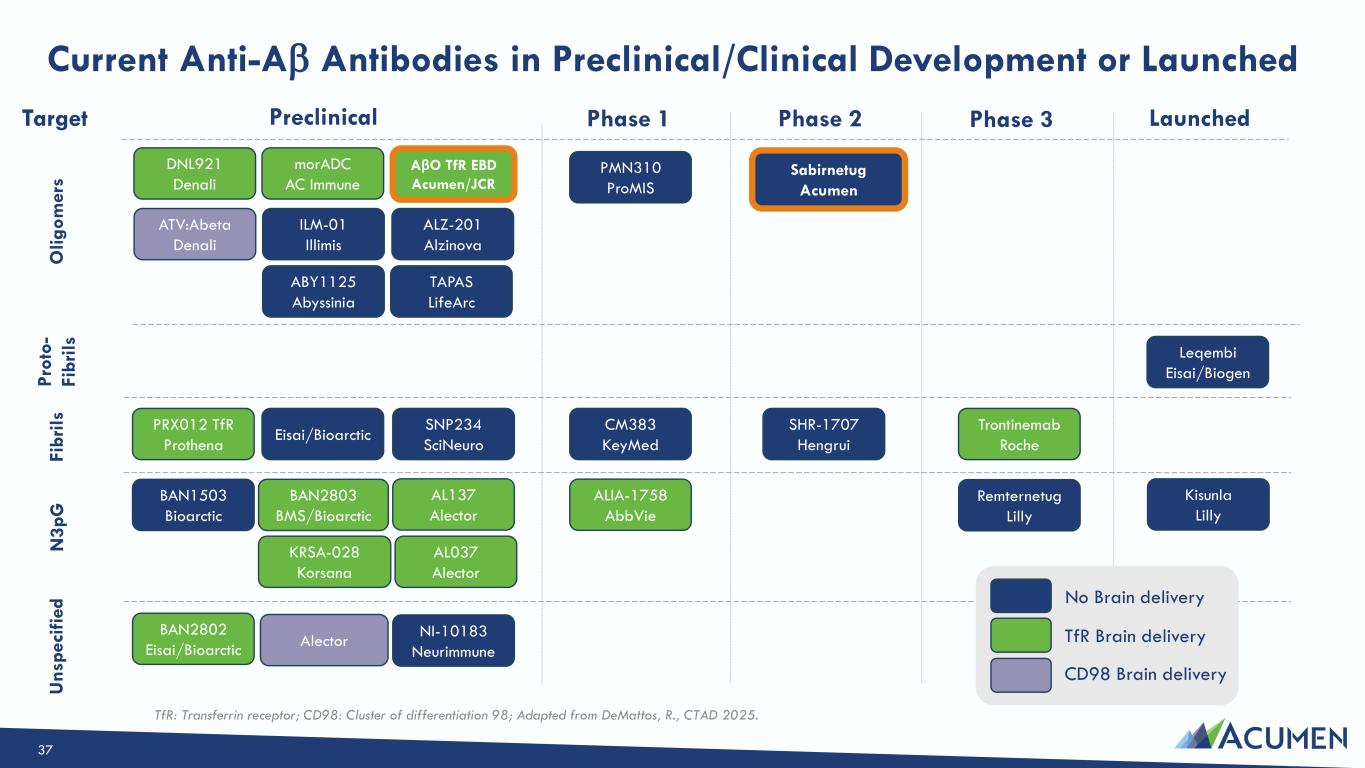
Current Anti-A Antibodies in Preclinical/Clinical Development or Launched O li g o m e rs Target Preclinical Phase 1 Phase 2 Phase 3 Launched Sabirnetug Acumen PMN310 ProMIS CM383 KeyMed Trontinemab Roche Leqembi Eisai/Biogen Kisunla Lilly Remternetug Lilly ALIA-1758 AbbVie TAPAS LifeArc ILM-01 Illimis ABY1125 Abyssinia AβO TfR EBD Acumen/JCR ALZ-201 Alzinova morADC AC Immune DNL921 Denali ATV:Abeta Denali BAN2802 Eisai/Bioarctic SNP234 SciNeuro BAN1503 Bioarctic AL137 Alector AL037 Alector Eisai/Bioarctic PRX012 TfR Prothena BAN2803 BMS/Bioarctic TfR Brain deliveryAlector CD98 Brain delivery TfR: Transferrin receptor; CD98: Cluster of differentiation 98; Adapted from DeMattos, R., CTAD 2025. F ib ri ls N 3 p G U n sp e ci fi e d SHR-1707 Hengrui NI-10183 Neurimmune No Brain delivery P ro to - F ib ri ls KRSA-028 Korsana 37

Non-clinical Sabirnetug Data
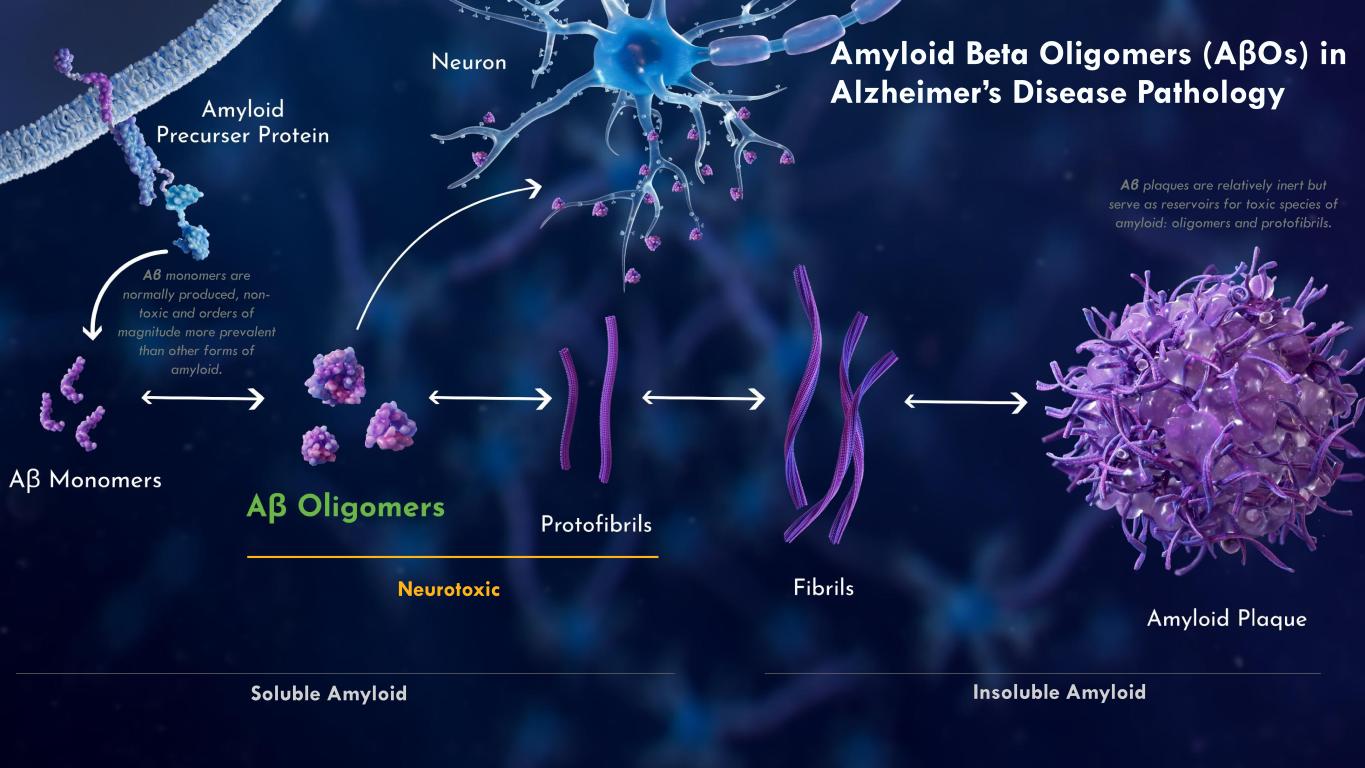
Amyloid Beta Oligomers (AβOs) in Alzheimer’s Disease Pathology Aβ monomers are normally produced, non- toxic and orders of magnitude more prevalent than other forms of amyloid. Aβ plaques are relatively inert but serve as reservoirs for toxic species of amyloid: oligomers and protofibrils. Neurotoxic Soluble Amyloid Insoluble Amyloid

Literature Selection: Soluble AβOs Contribute to Pathophysiological Processes Associated with Alzheimer’s Disease Disrupted Ca2+ homeostasis Demuro et al, 2005 De Felice et al, 2007 Alberdi et al, 2010 Wang et al, 2018 ChAT loss Heinitz et al, 2006 Nunes-Tavares et al, 2012 Inhibition of axonal transport Pigino et al, 2009 Poon et al, 2009 Decker et al, 2010 Oxidative stress Longo et al, 2000 Sponne et al, 2003 Tabner et al, 2005 De Felice et al, 2007 ER stress Resende et al, 2008 Nishitsuji et al, 2009 Insulin resistance Zhao et al, 2008 Zhao et al, 2009 Ma et al, 2009 De Felice et al, 2009 Impact on astrocytes/microglia Hu et al, 1998 Jimenez et al, 2008 Sondag et al, 2009 Tomiyama et al, 2010 Aberrant Tau phosphorylation De Felice et al, 2008 Ma et al, 2009 Tomiyama et al, 2010 Zempel et al, 2010 Bloom, 2014 Forny-Germano et al, 2020 Wakeman et al, 2022 Darricau et al, 2023 Selective neuron death Lambert et al, 1998 Kim et al, 2003 Florent et al, 2006 Ryan et al, 2009 Lee et al, 2017 Komura, 2019 Plasticity dysfunction Lambert et al, 1998 Walsh et al, 2002 Wang et al, 2002 Townsend et al, 2006 Yasumoto et al, 2019 Synapse deterioration Zhao et al, 2006 Lacor et al, 2007 Shankar et al, 2007 Wu et al, 2010 Brito-Moreira et al, 2017 Actor-Engel et al, 2021 Sackmann & Hallbeck, 2020 Limegrover et al, 2021 Receptor Redistribution Snyder et al, 2005 Roselli et al, 2005 Lacor et al, 2007 Zhao et al, 2008 40
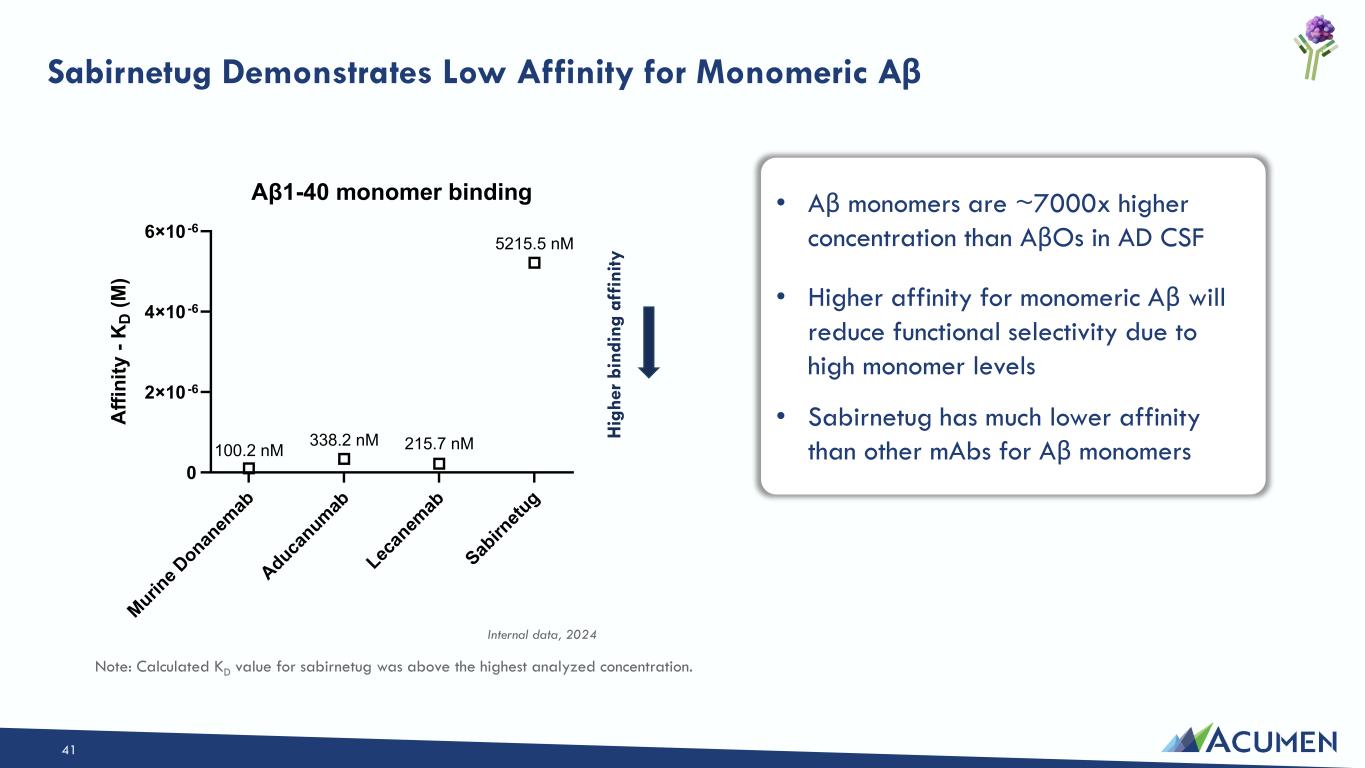
• Aβ monomers are ~7000x higher concentration than AβOs in AD CSF • Higher affinity for monomeric Aβ will reduce functional selectivity due to high monomer levels • Sabirnetug has much lower affinity than other mAbs for Aβ monomers Sabirnetug Demonstrates Low Affinity for Monomeric Aβ 41 M urin e D onan em ab A duca num ab Lec an em ab S ab irn et ug 0 2×10 -6 4×10 -6 6×10 -6 Aβ1-40 monomer binding A ff in it y - K D ( M ) 100.2 nM 338.2 nM 215.7 nM 5215.5 nM H ig h e r b in d in g a ff in it y Internal data, 2024 Note: Calculated KD value for sabirnetug was above the highest analyzed concentration.

Sabirnetug is Highly Selective for Aβ Oligomers Versus Aβ Monomers Relative Selectivity for AβO versus Monomeric Aβ Measured with SPR 42 Sabirnetug is more selective for AβOs than aducanumab or lecanemab Aducanumab Lecanemab Sabirnetug 1×10 -10 1×10 -9 1×10 -8 1×10 -7 1×10 -6 1×10 -5 Sabirnetug vs Lecanemab and Aducanumab A ff in it y - K D ( M ) Aβ1-40 monomer Aβ oligomers 8750x 222x 79.6x H ig h e r b in d in g a ff in it y Internal data, 2024 Note: Murine donanemab shows very low signals for Aβ oligomer binding compared to all other antibodies tested; therefore, it was not included in this comparison.
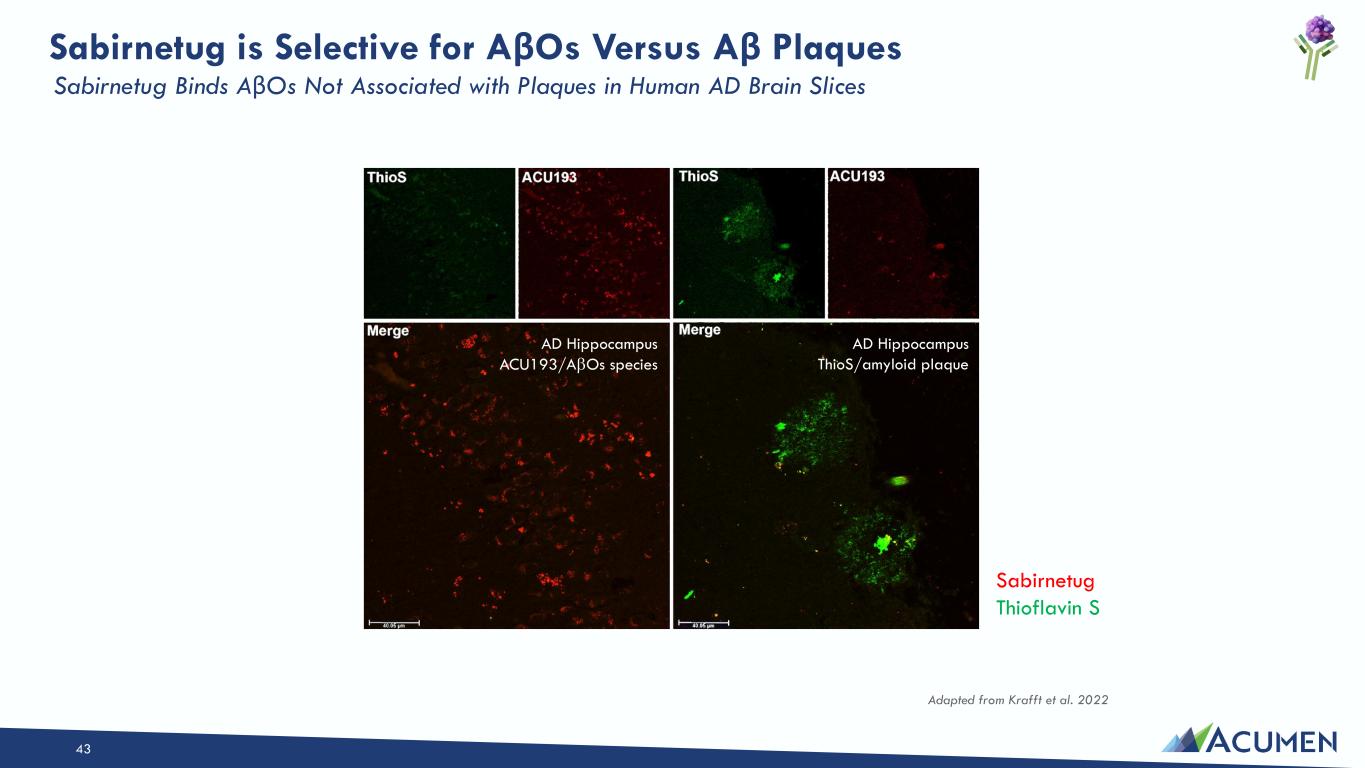
43 AD Hippocampus ThioS/amyloid plaque AD Hippocampus ACU193/AOs species Sabirnetug is Selective for AβOs Versus Aβ Plaques Sabirnetug Thioflavin S Sabirnetug Binds AβOs Not Associated with Plaques in Human AD Brain Slices Adapted from Krafft et al. 2022
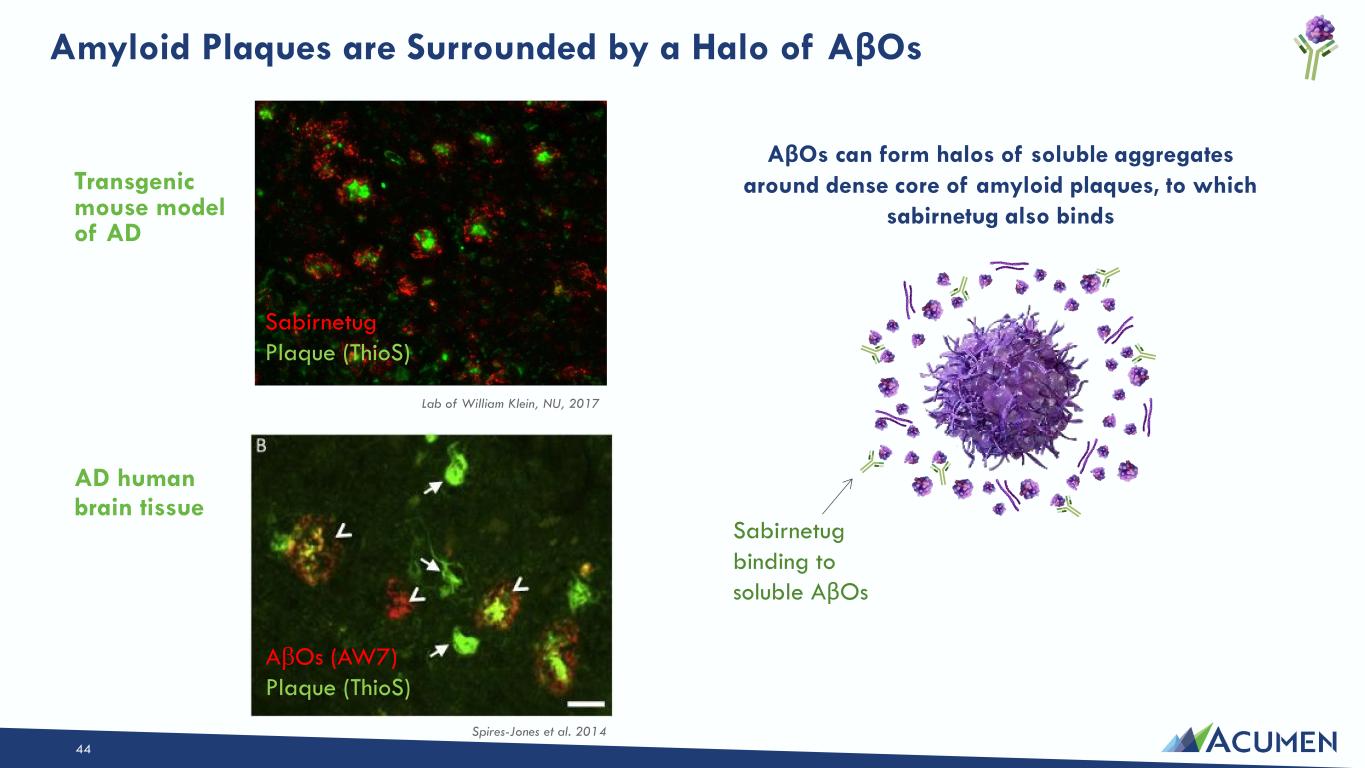
44 Amyloid Plaques are Surrounded by a Halo of AβOs Transgenic mouse model of AD AβOs can form halos of soluble aggregates around dense core of amyloid plaques, to which sabirnetug also binds Sabirnetug binding to soluble AβOs Sabirnetug Plaque (ThioS) Spires-Jones et al. 2014 AOs (AW7) Plaque (ThioS) AD human brain tissue Lab of William Klein, NU, 2017

Sabirnetug: Extensive Data Package Supporting Development Sabirnetug is a promising immunotherapy for early AD expected to provide meaningful cognitive and functional benefits, slow disease progression, and offer an attractive safety profile Nanomolar affinity for AβOs, >500-fold greater selectivity for AβOs over Aβ monomer, with limited or no discernable binding to vascular amyloid or dense core amyloid plaques Binds broad range of endogenous Aβ, from dimers to high molecular weight AβOs Brain penetration and biodistribution demonstrated in multiple species Performs like other peripherally administered CNS mAbs Dose-dependent effects in multiple in vitro neuroprotection assays Positive memory and behavioral effects in multiple in vivo transgenic mouse models for AD IgG2 subclass lacks inflammatory effector function signaling (FcγR binding) Non-clinical microhemorrhage studies show no increased risk of microhemorrhage GLP studies demonstrated acceptable safety package supporting clinical dosing plans including Phase 2 SELECTIVITY PHARMACOLOGY PK/PD SAFETY 45
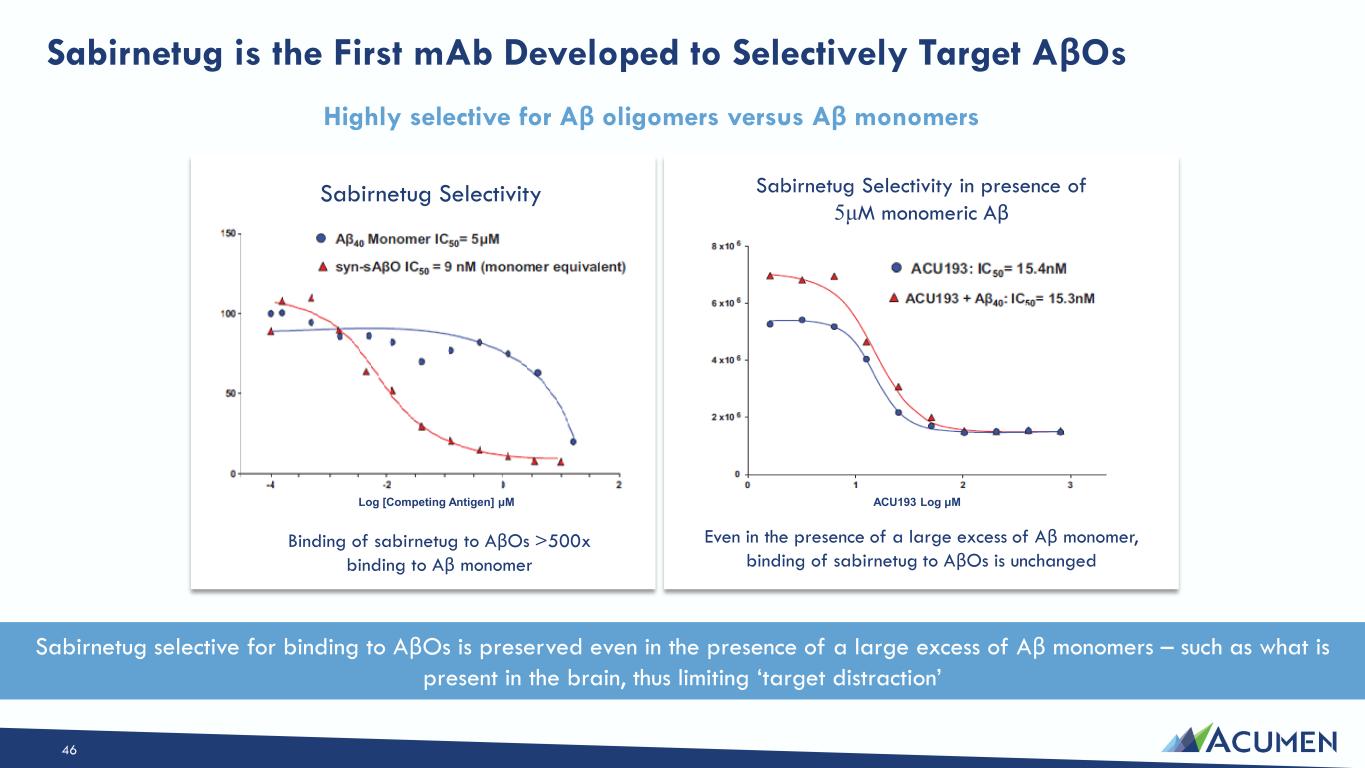
Highly selective for Aβ oligomers versus Aβ monomers Even in the presence of a large excess of Aβ monomer, binding of sabirnetug to AβOs is unchanged Binding of sabirnetug to AβOs >500x binding to Aβ monomer Log [Competing Antigen] μM Sabirnetug Selectivity Sabirnetug Selectivity in presence of 5μM monomeric Aβ ACU193 Log μM Sabirnetug is the First mAb Developed to Selectively Target AβOs Sabirnetug selective for binding to AβOs is preserved even in the presence of a large excess of Aβ monomers – such as what is present in the brain, thus limiting ‘target distraction’ 46
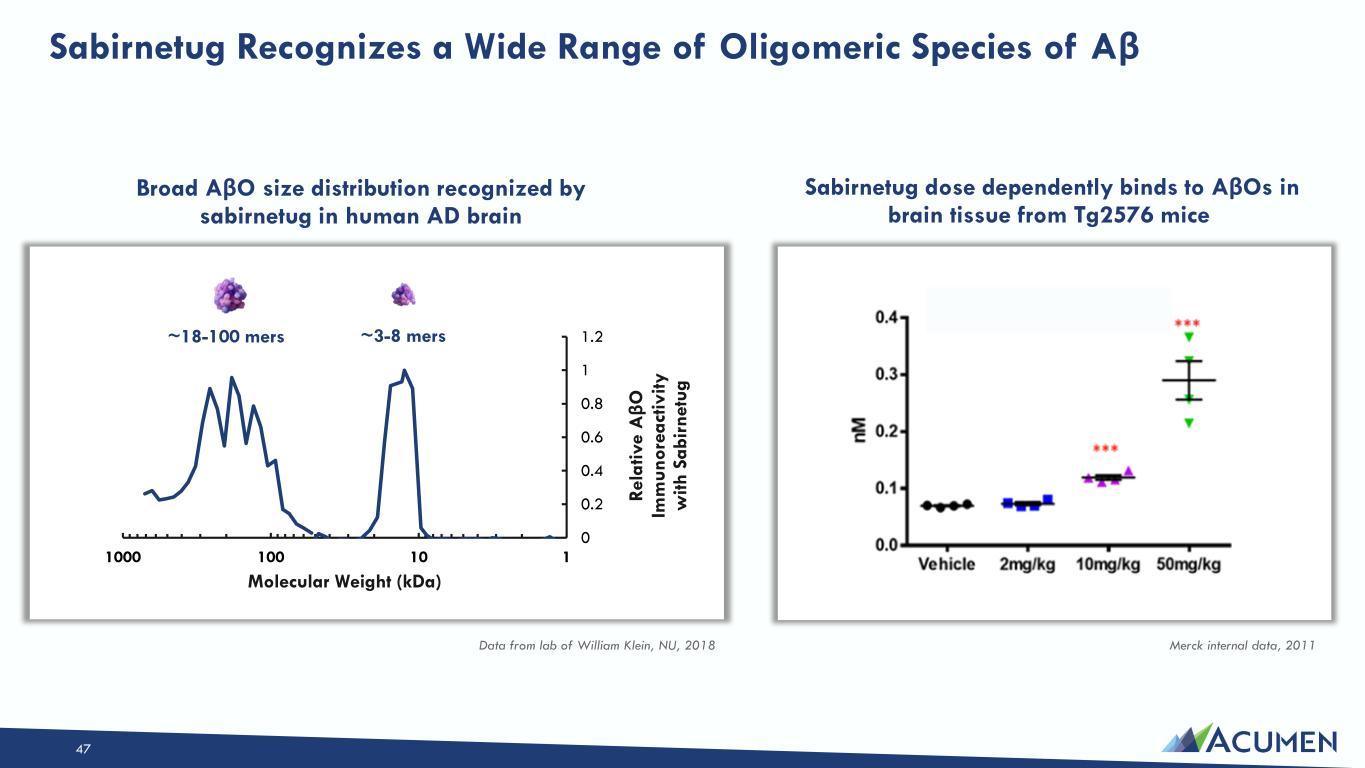
47 Sabirnetug Recognizes a Wide Range of Oligomeric Species of Aβ Broad AβO size distribution recognized by sabirnetug in human AD brain Sabirnetug dose dependently binds to AβOs in brain tissue from Tg2576 mice Data from lab of William Klein, NU, 2018 Merck internal data, 2011 0 0.2 0.4 0.6 0.8 1 1.2 1101001000 R e la ti v e A β O Im m u n o re a ct iv it y w it h S a b ir n e tu g Molecular Weight (kDa) ~3-8 mers~18-100 mers
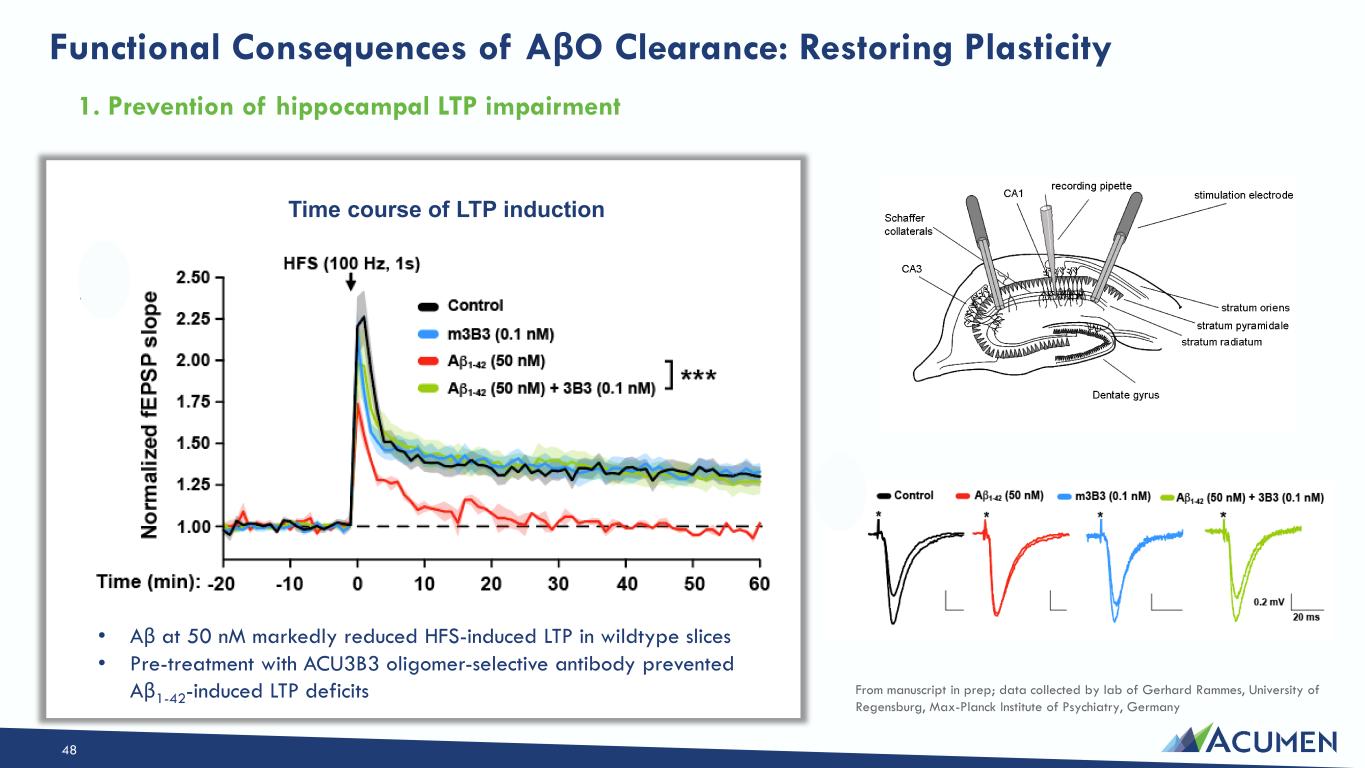
1. Prevention of hippocampal LTP impairment Functional Consequences of AβO Clearance: Restoring Plasticity 48 • Aβ at 50 nM markedly reduced HFS-induced LTP in wildtype slices • Pre-treatment with ACU3B3 oligomer-selective antibody prevented Aβ1-42-induced LTP deficits Time course of LTP induction From manuscript in prep; data collected by lab of Gerhard Rammes, University of Regensburg, Max-Planck Institute of Psychiatry, Germany
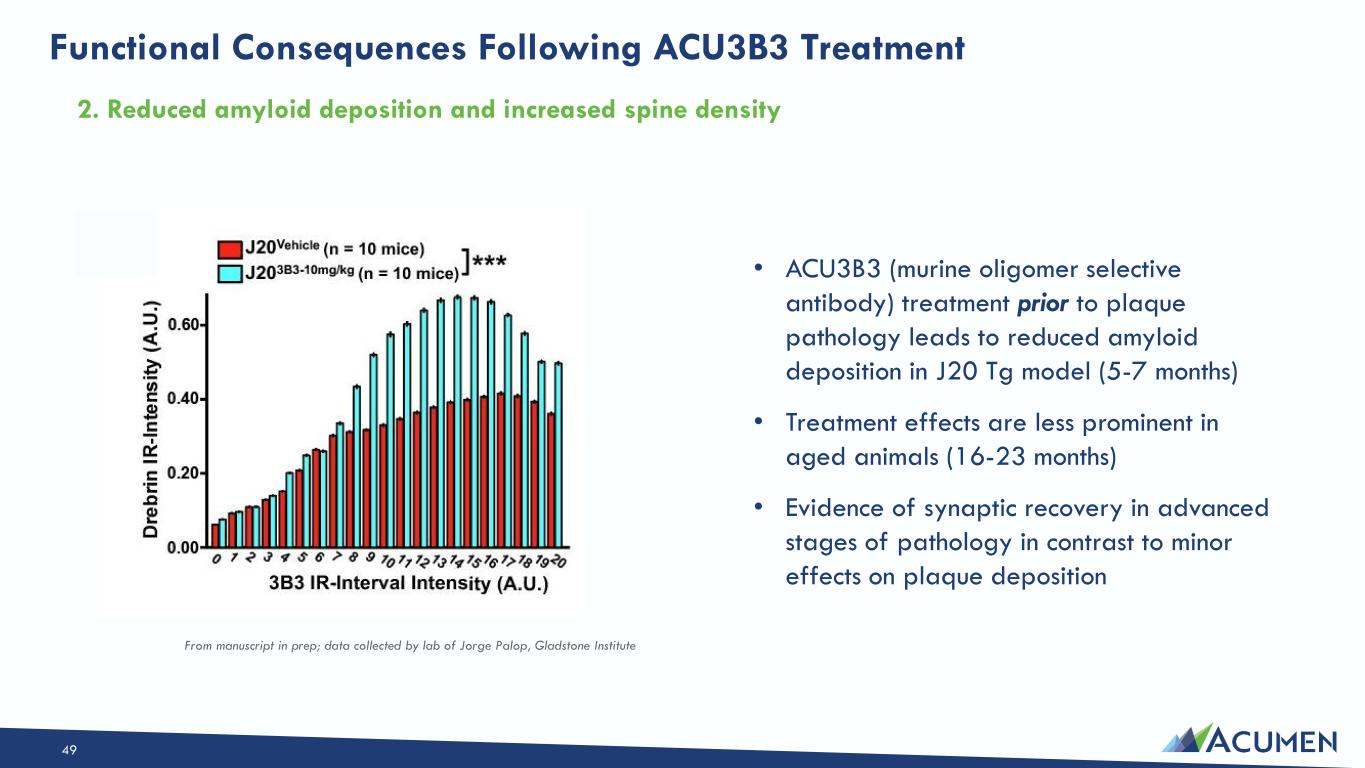
Functional Consequences Following ACU3B3 Treatment 49 • ACU3B3 (murine oligomer selective antibody) treatment prior to plaque pathology leads to reduced amyloid deposition in J20 Tg model (5-7 months) • Treatment effects are less prominent in aged animals (16-23 months) • Evidence of synaptic recovery in advanced stages of pathology in contrast to minor effects on plaque deposition From manuscript in prep; data collected by lab of Jorge Palop, Gladstone Institute 2. Reduced amyloid deposition and increased spine density

Phase 1 INTERCEPT-AD
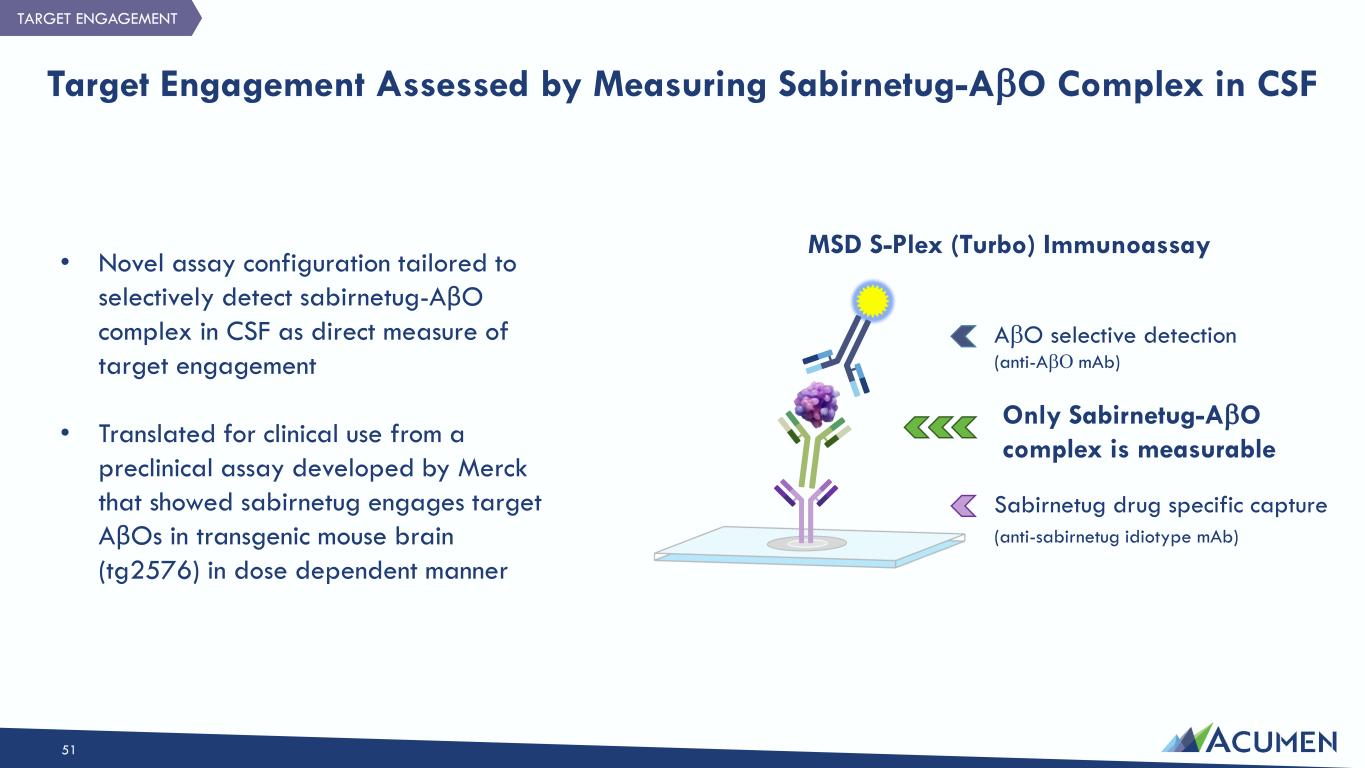
Sabirnetug drug specific capture (anti-sabirnetug idiotype mAb) 51 Target Engagement Assessed by Measuring Sabirnetug-AO Complex in CSF AO selective detection (anti-A mAb) Only Sabirnetug-AO complex is measurable • Novel assay configuration tailored to selectively detect sabirnetug-AβO complex in CSF as direct measure of target engagement • Translated for clinical use from a preclinical assay developed by Merck that showed sabirnetug engages target AβOs in transgenic mouse brain (tg2576) in dose dependent manner MSD S-Plex (Turbo) Immunoassay TARGET ENGAGEMENT
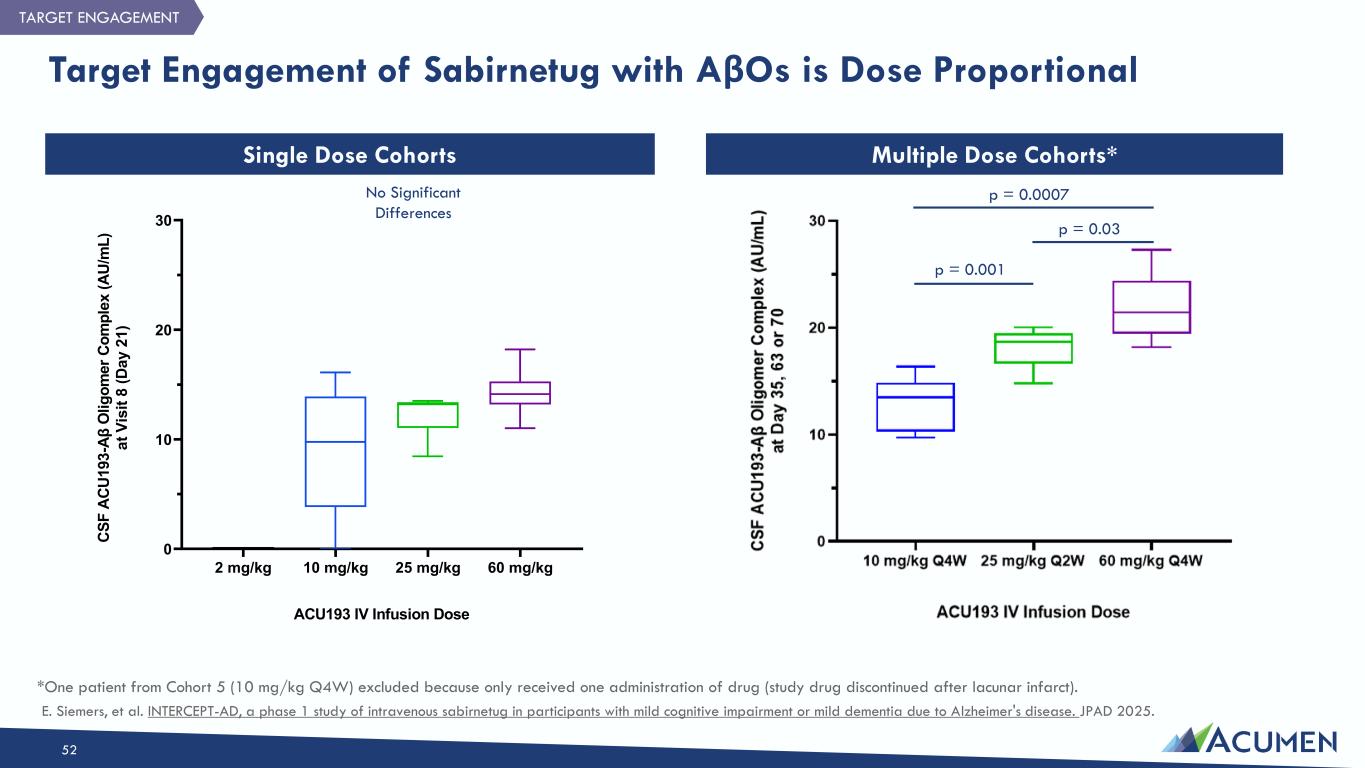
Target Engagement of Sabirnetug with AβOs is Dose Proportional 52 Single Dose Cohorts Multiple Dose Cohorts* 2 mg/kg 10 mg/kg 25 mg/kg 60 mg/kg 0 10 20 30 ACU193 IV Infusion Dose C S F A C U 1 9 3 -A β O li g o m e r C o m p le x ( A U /m L ) a t V is it 8 ( D a y 2 1 ) *One patient from Cohort 5 (10 mg/kg Q4W) excluded because only received one administration of drug (study drug discontinued after lacunar infarct). TARGET ENGAGEMENT p = 0.001 p = 0.03 p = 0.0007No Significant Differences E. Siemers, et al. INTERCEPT-AD, a phase 1 study of intravenous sabirnetug in participants with mild cognitive impairment or mild dementia due to Alzheimer's disease. JPAD 2025.
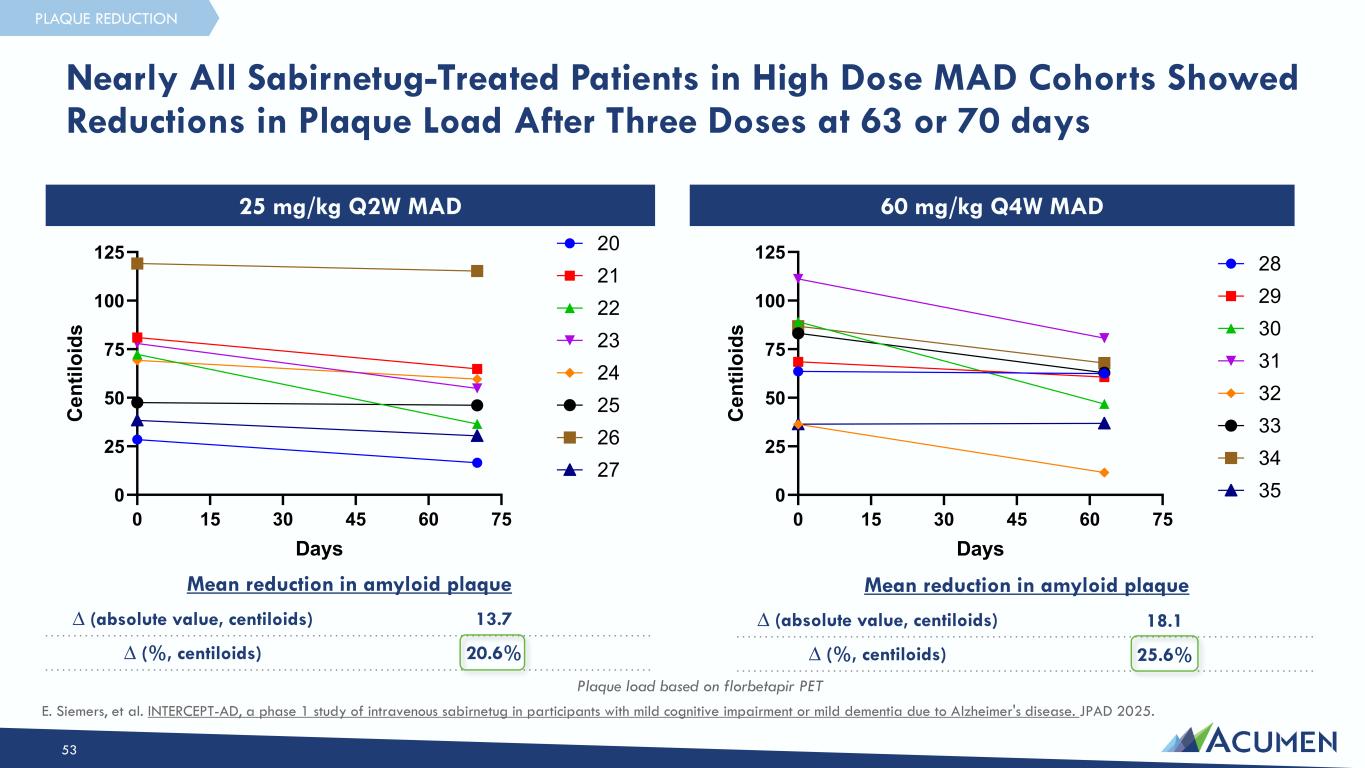
Nearly All Sabirnetug-Treated Patients in High Dose MAD Cohorts Showed Reductions in Plaque Load After Three Doses at 63 or 70 days 53 Plaque load based on florbetapir PET 0 15 30 45 60 75 0 25 50 75 100 125 25 mg/kg Q2W MAD Days C e n ti lo id s 20 21 22 23 24 25 26 27 0 15 30 45 60 75 0 25 50 75 100 125 60 mg/kg Q4W MAD Days C e n ti lo id s 28 29 30 31 32 33 34 35 25 mg/kg Q2W MAD 60 mg/kg Q4W MAD Mean reduction in amyloid plaque ∆ (absolute value, centiloids) 13.7 ∆ (%, centiloids) 20.6% Mean reduction in amyloid plaque ∆ (absolute value, centiloids) 18.1 ∆ (%, centiloids) 25.6% PLAQUE REDUCTION E. Siemers, et al. INTERCEPT-AD, a phase 1 study of intravenous sabirnetug in participants with mild cognitive impairment or mild dementia due to Alzheimer's disease. JPAD 2025.
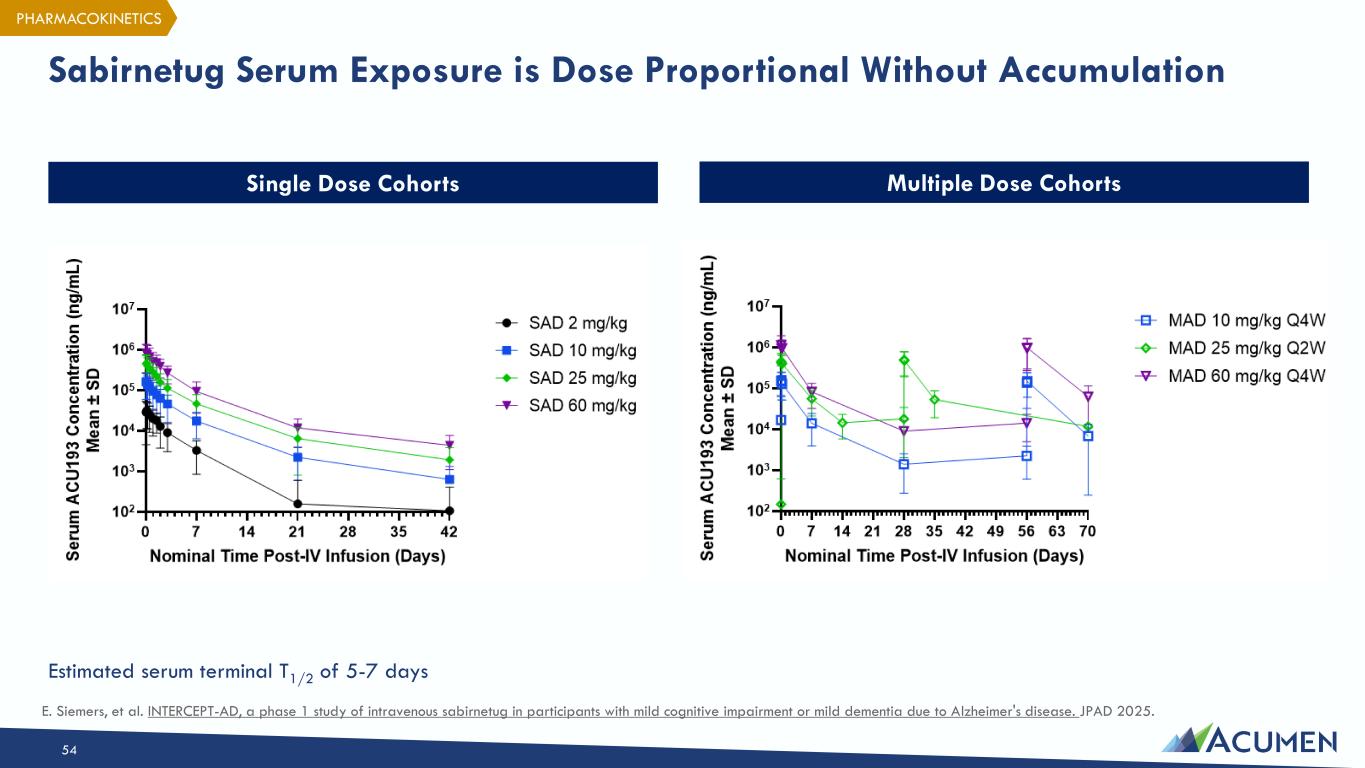
Sabirnetug Serum Exposure is Dose Proportional Without Accumulation 54 Single Dose Cohorts Multiple Dose Cohorts PHARMACOKINETICS Estimated serum terminal T1/2 of 5-7 days E. Siemers, et al. INTERCEPT-AD, a phase 1 study of intravenous sabirnetug in participants with mild cognitive impairment or mild dementia due to Alzheimer's disease. JPAD 2025.
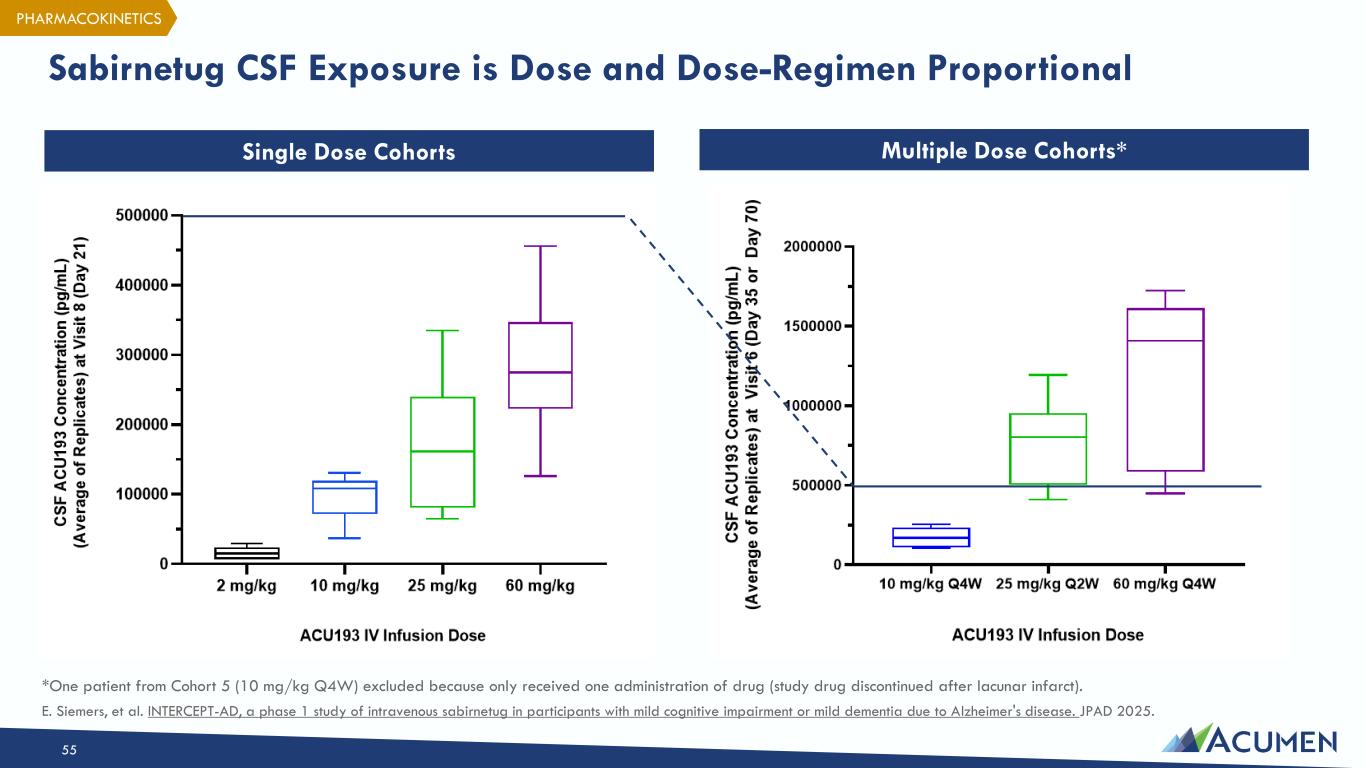
Sabirnetug CSF Exposure is Dose and Dose-Regimen Proportional 55 Single Dose Cohorts Multiple Dose Cohorts* *One patient from Cohort 5 (10 mg/kg Q4W) excluded because only received one administration of drug (study drug discontinued after lacunar infarct). PHARMACOKINETICS E. Siemers, et al. INTERCEPT-AD, a phase 1 study of intravenous sabirnetug in participants with mild cognitive impairment or mild dementia due to Alzheimer's disease. JPAD 2025.
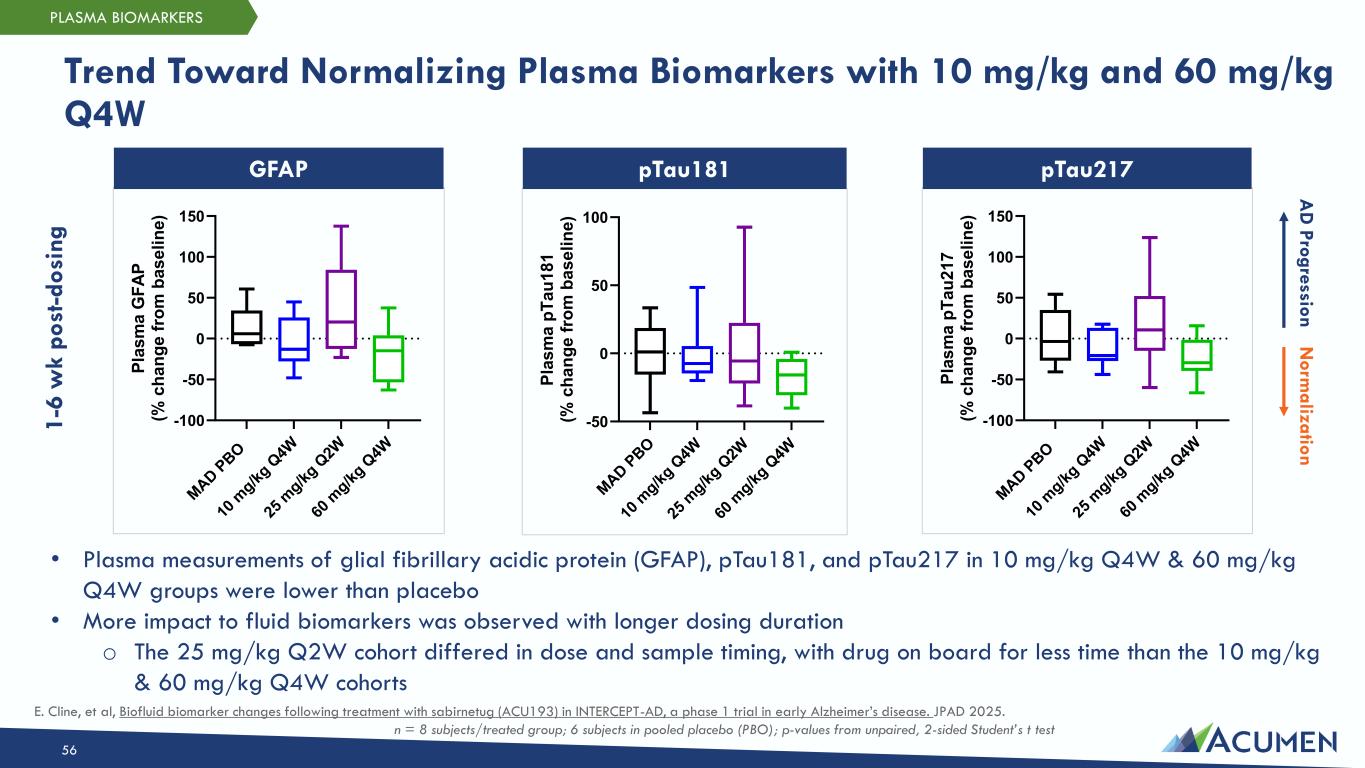
Trend Toward Normalizing Plasma Biomarkers with 10 mg/kg and 60 mg/kg Q4W 56 M A D P B O 10 m g/k g Q 4W 25 m g/k g Q 2W 60 m g/k g Q 4W -100 -50 0 50 100 150 P la s m a G F A P (% c h a n g e f ro m b a s e li n e ) M A D P B O 10 m g/k g Q 4W 25 m g/k g Q 2W 60 m g/k g Q 4W -50 0 50 100 P la s m a p T a u 1 8 1 (% c h a n g e f ro m b a s e li n e ) M A D P B O 10 m g/k g Q 4W 25 m g/k g Q 2W 60 m g/k g Q 4W -100 -50 0 50 100 150 P la s m a p T a u 2 1 7 (% c h a n g e f ro m b a s e li n e ) 1 -6 w k p o st -d o si n g • Plasma measurements of glial fibrillary acidic protein (GFAP), pTau181, and pTau217 in 10 mg/kg Q4W & 60 mg/kg Q4W groups were lower than placebo • More impact to fluid biomarkers was observed with longer dosing duration o The 25 mg/kg Q2W cohort differed in dose and sample timing, with drug on board for less time than the 10 mg/kg & 60 mg/kg Q4W cohorts PLASMA BIOMARKERS pTau181GFAP pTau217 A D P ro g re ssio n N o rm a liz a tio n n = 8 subjects/treated group; 6 subjects in pooled placebo (PBO); p-values from unpaired, 2-sided Student’s t test E. Cline, et al, Biofluid biomarker changes following treatment with sabirnetug (ACU193) in INTERCEPT-AD, a phase 1 trial in early Alzheimer’s disease. JPAD 2025.

Phase 2 ALTITUDE-AD
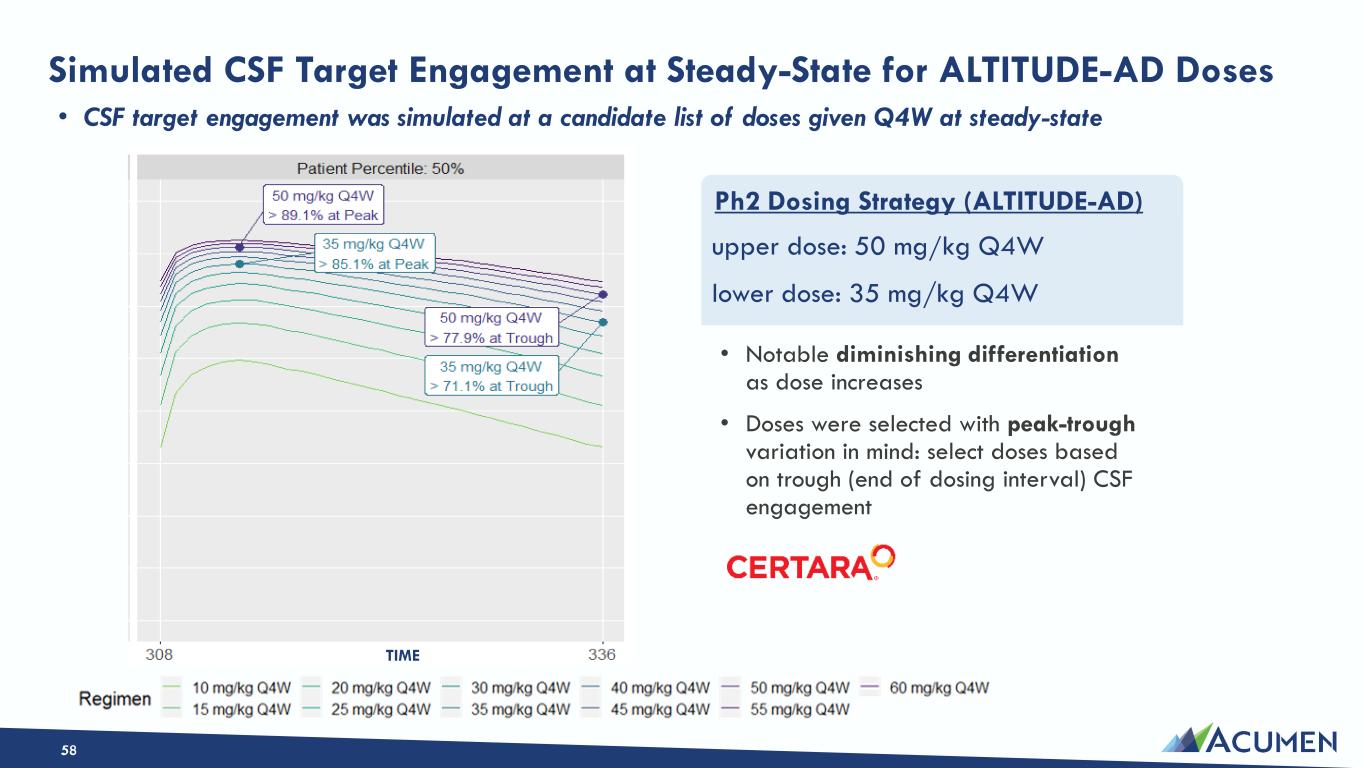
Simulated CSF Target Engagement at Steady-State for ALTITUDE-AD Doses • CSF target engagement was simulated at a candidate list of doses given Q4W at steady-state • Notable diminishing differentiation as dose increases • Doses were selected with peak-trough variation in mind: select doses based on trough (end of dosing interval) CSF engagement upper dose: 50 mg/kg Q4W lower dose: 35 mg/kg Q4W Ph2 Dosing Strategy (ALTITUDE-AD) TIME 58
